Jobs
Chemistry Notes – Acid, Bases and Salts
Published
7 years agoon
[ad_1]
Click Here – Free KCSE Past Papers » KNEC Past Exams » Free Downloads » KCSE Papers & Marking Schemes
K.C.S.E Online Revision
A. Acids And Bases
At a school laboratory:
(i)An acid may be defined as a substance that turn litmus red.
(ii)A base may be defined as a substance that turn litmus blue.
Litmus is a lichen found mainly in West Africa. It changes its colour depending on
whether the solution it is in, is basic/alkaline or acidic.It is thus able to
identify/ show whether
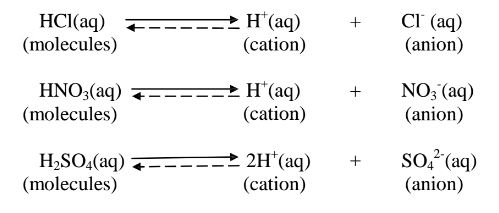
1. An acid is a substance that dissolves in water to form H+/H3O+ as the only
positive ion/cation. This is called the Arrhenius definition of an acid. From this
definition, an acid dissociate/ionize in water releasing H+ thus:

2.A base is a substance which dissolves in water to form OH‘ as the only
negatively charged ion/anion.
This is called Arrhenius definition of a base.
From this definition, a base dissociate/ionize in Water releasing OH’ thus:

3. An acid is a proton donor.
A base is a proton acceptor.
This is called Bronsted-Lowry definition of acids and bases.
From this definition, an acid donates H+ .
H+ has no electrons and neutrons .It contains only a proton.
Examples
i. From the equation:
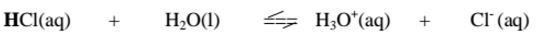
(a)(i)For the forward reaction from left to right, H2O gains a proton to form
H3O+ and thus H2o is a proton acceptor.It is a Bronsted-Lowry base
(ii) For the backward reaction from right to left, H3O+ donates a proton to
form H2o and thus H3O+ is an ,,opposite” proton donor. It is a Bronsted-
Lowry conjugate acid
(b)(i)For the forward reaction from left to right, HCl donates a proton to
form Cl‘ and thus HCl is a proton donor.
It is a Bronsted-Lowry acid
(ii) For the backward reaction from right to left, Cl” gains a proton to form
HCl and thus Cl’ is an ,,opposite” proton acceptor.
It is a Bronsted-Lowry conjugate base.
Every base /acid from Bronsted-Lowry definition thus must have a conjugate
product/reactant.
II. From the equation:
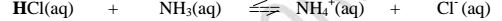
(a)(i)For the forward reaction from left to right, NH; gains a proton to form
NH4 and thus NH3; is a proton acceptor .
It is a Bronsted-Lowry base
(ii) For the backward reaction from right to left, NH4+ donates a proton to
form NH; and thus NH4+ is an ,,opposite” proton donor.
It is a Bronsted-Lowry conjugate acid
(b)(i)F or the forward reaction from left to right, HCl donates a proton to
form C1‘ and thus HCl is a proton donor .
It is a Bronsted-Lowry acid
(ii) For the backward reaction from right to left, Cl” gains a proton to form
HCl and thus Cl’ is an ,,opposite” proton acceptor.
It is a Bronsted-Lowry conjugate base.
4. Acids and bases show acidic and alkaline properties/characteristics only in
Water but not in other solvents e.g.
(a)Hydrogen chloride gas dissolves in water to form hydrochloric acid
Hydrochloric acid dissociates/ionizes in water to free
ions. The free
ions are responsible for:
(i)turning blue litmus paper/solution red.
(ii)show pH value 1/2/3/4/5/6
(iii)are good electrolytes/conductors of electricity/undergo electrolysis.
(iv)react with metals to produce /evolve hydrogen gas and a salt. i.e.
Ionically:

(v) react with metal carbonates and hydrogen carbonates to produce /evolve
carbon(IV)oxide gas ,Water and a salt. i.e.
lonically:

(vi)neutralize metal oxides/hydroxides to salt and water only. i.e.
Ionically:

(b)Hydrogen chloride gas dissolves in methylbenzene /benzene but does not
dissociate /ionize into free ions.
It exists in molecular state showing none of the above properties.
(c)Ammonia gas dissolves in water to form aqueous ammonia which
dissociate/ionize to free NH4+ (aq) and OH'(aq) ions.
This dissociation/ionization makes aqueous ammonia to:
(i)tum litmus paper/solution blue.
(ii)have pH 8/9/ 10/ 11
(iii)be a good electrical conductor
(iv)react with acids to form ammonium salt and water only.
(d)Ammonia gas dissolves in methylbenzene/benzene /kerosene but does not
dissociate into free ions therefore existing as molecules
6. Solvents are either polar or non-polar.
A polar solvent is one which dissolves ionic compounds and other polar solvents.
Water is polar solvent that dissolves ionic and polar substance by surrounding the
free ions as below:
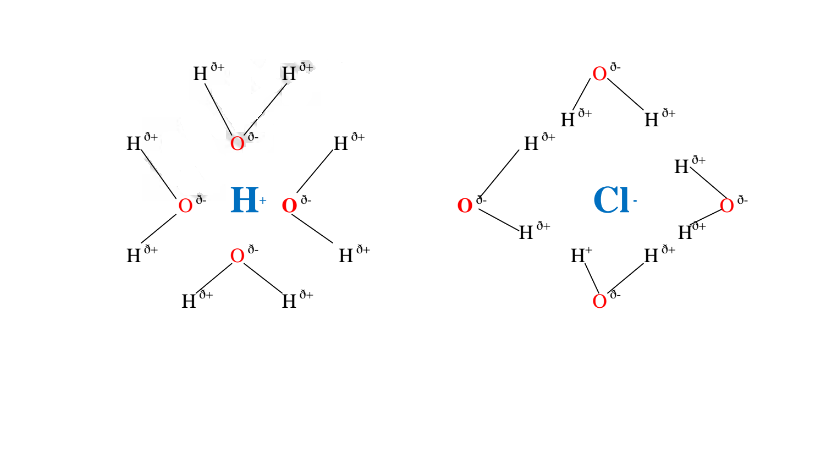
Note:Water is polar .It is made up of :
Oxygen atom is partially negative and two hydrogen atoms which are partially
positive.
They surround the free Hl and Cl” ions.
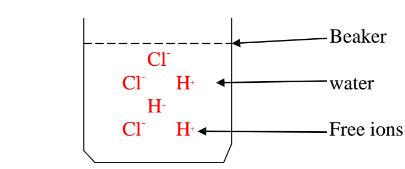
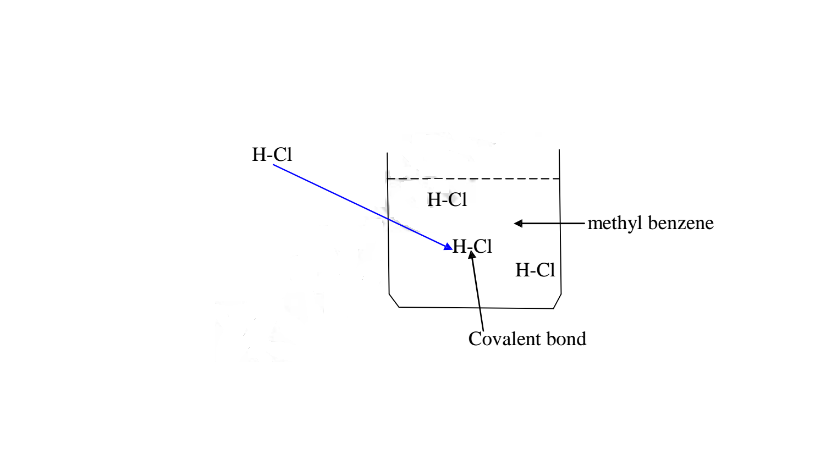
A non polar solvent is one which dissolved non-polar substances and covalent
compounds.
If a polar ionic compound is dissolved in non-polar solvent ,it does not
ionize/dissociate into free ions as below:
7. Some acids and bases are strong while others are Weak.
(a)A strong acid/base is one which is fully/wholly/completely dissociated / ionized
into many free H+ /OH’ ions i.e.
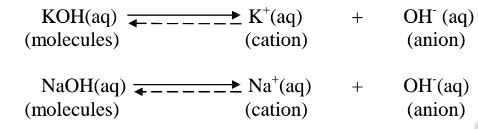
i. Strong acids exists more as free H+ ions than molecules. e. g.
ii. Strong bases/alkalis exists more as free OH’ ions than molecules. e.g.
(b) A weak base/acid is one which is paitially /partly dissociated /ionized in water
into free OH’ (aq) and H+(aq) ions.
i. Weak acids exists more as molecules than as free Hl ions. e.g.
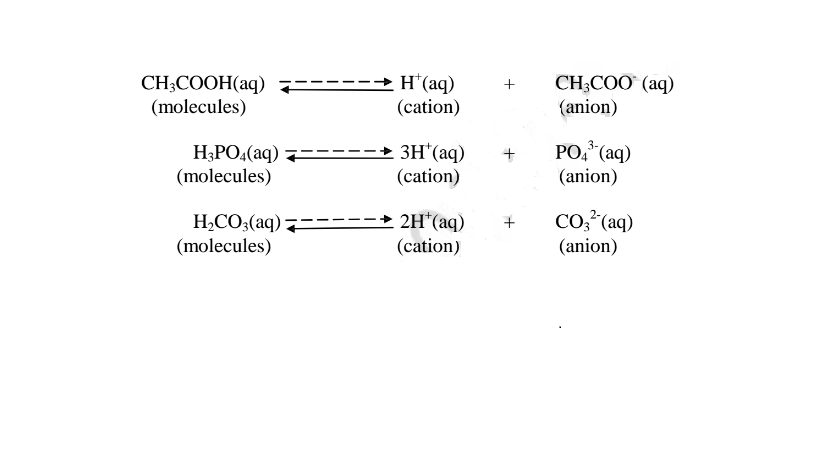
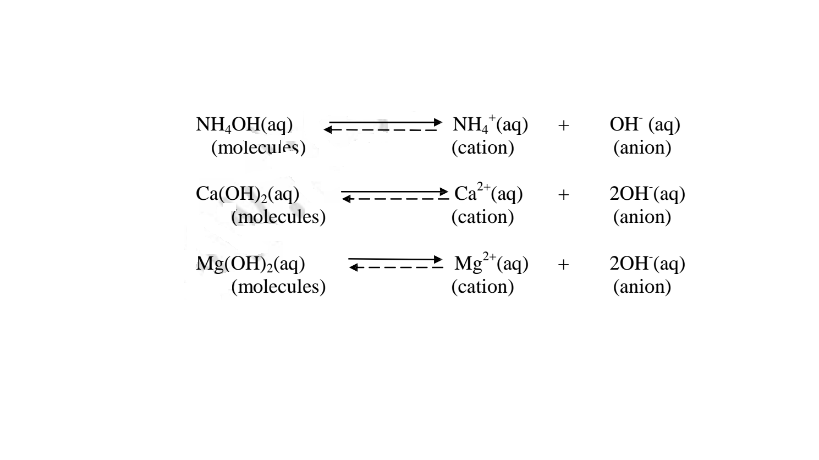
ii. Weak bases/alkalis exists more as molecules than free OH’ ions. e. g.
8. The concentration of an acid/base/alkali is based on the number of moles of
acid/bases dissolved in a decimeter(litre)of the solution.
An acid/base/alkali With more acid/base/alkali in a decimeter(litre) of solution is
said to be concentrated while that with less is said to be dilute.
9. (a) (i)strong acids have pH 1/2/3 while weak acids have high pH 4/5/6.
(ii)a neutral solution have pH 7
(iii)strong alkalis/bases have pH 12/13/14 while weak bases/alkalis have pH
11/10 /9 / 8.
(b) pH is a measure of H+(aq) concentration in a solution.
The higher the H+(aq)ions concentration ;
-the higher the acidity
-the lower the pH
-the lower the concentration of OH_(aq)
-the lower the alkalinity
At pH 7 , a solution has equal concentration of H+(aq) and OH'(aq).
Beyond pH 7,the concentration of the OH'(aq) increases as the H+(aq) ions
decreases.
10.(a) When acids /bases dissolve in water, the ions present in the solution conduct
electricity.
The more the dissociation the higher the yield of ions and the greater the electrical
conductivity of the solution.
A compound that conducts electricity in an electrolyte and thus a compound
showing high electrical conductivity is a strong electrolyte while a compound
showing low electrical conductivity is a weak electrolyte.
(b) Practically, a bright light on a bulb ,a high voltage reading from a voltmeter
high ammeter reading from an ammeter, a big deflection on a galvanometer is an
indicator of strong electrolyte(acid/base) and the opposite for weak
electrolytes(acids/base)
11. Some compounds exhibit/show both properties of acids and bases/alkalis.
A substance that reacts with both acids and bases is said to be amphotellic.
The examples below show the amphotellic properties of:
(a)
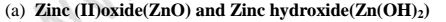
(i)When 1/2 spatula full of Zinc(II)oxide is placed in a boiling tube containing
10cm3 of either 2M nitric(V)acid or 2M sodium hydroxide hydroxide solution, it
dissolves on both the acid and the alkali/base to form a colourless solution. i.e.
(i) when reacting with nitric(V)acid, the oxide shows basic properties by
reacting with an acid to form a simple salt and water only.
Basic oxide + Acid -> salt + water
Examples:

(ii) when reacting with sodium hydroxide, the oxide shows acidic properties by
reacting with a base to form a complex salt.
Basic oxide + Base/alkali + Water -> Complex salt
Examples:
Chemical equation
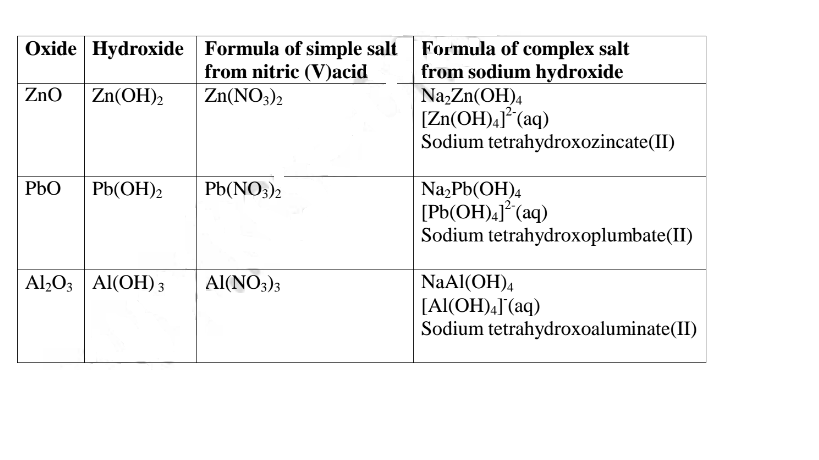
1.When Zinc oxide is reacted with sodium hydroxide the complex salt is sodium
tetrahydroxozincate(II) complex salt.
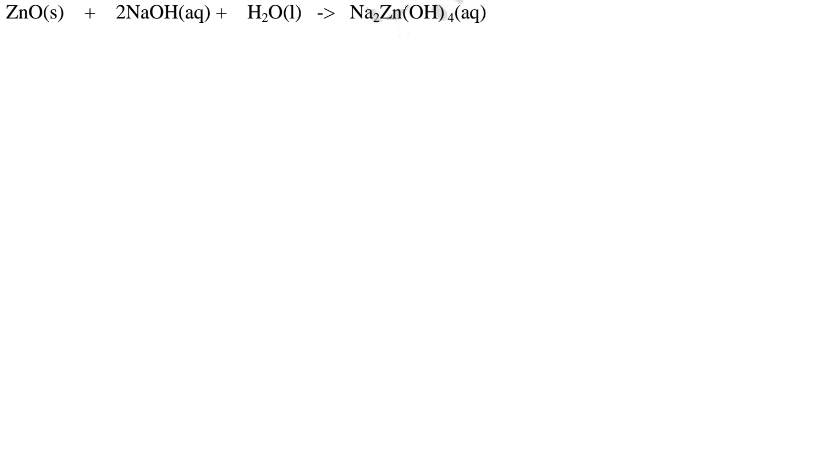
2.When Zinc oxide is reacted with potassium hydroxide the complex salt is
potassium tetrahydroxozincate(II) complex salt.

(ii)When Zinc(ll)hydroxide is placed in a boiling tube containing 1Ocm3 of either
2M nitric(V)acid or 2M sodium hydroxide hydroxide solution, it dissolves on both
the acid and the alkali/base to form a colourless solution. i.e.
(i) when reacting with nitric(V)acid, the hydroxide shows basic properties. It
reacts with an acid to form a simple salt and water only.
Basic hydroxide + Acid -> salt + water
Examples:

(ii) when reacting with sodium hydroxide, the hydroxide shows acidic properties
by reacting with a base to form a complex salt.
Basic hydroxide + Base/alkali -> Complex salt
Examples:
Chemical equation
1.When Zinc hydroxide is reacted with sodium hydroxide the complex salt is
sodium tetrahydroxozineate(II) complex salt.
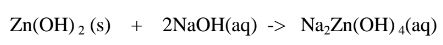
2.When Zinc hydroxide is reacted with potassium hydroxide the complex salt is
potassium tetrahydroxozincate(II) complex salt.

(b) Lead (II)oxide(PbO) and Lead(II) hydroxide (Pb(OH)2)
(i)When 1/2 spatula full of Lead(II)oxide is placed in a boiling tube containing
10cm3 of either 2M nitric(V)acid or 2M sodium hydroxide hydroxide solution, it
dissolves on both the acid and the alkali/base to form a colourless solution. i.e.
(i) when reacting with nitric(V)acid, the oxide shows basic properties by
reacting with an acid to form a simple salt and water only. All other Lead salts are
insoluble.

(ii) when reacting with sodium hydroxide, the oxide shows acidic properties by
reacting with a base to form a complex salt.
Chemical equation
1.When Lead(II) oxide is reacted with sodium hydroxide the complex salt is
sodium tetrahydroxoplumbate(II) complex salt.
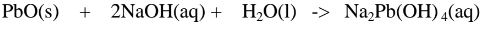
2.When Lead(II) oxide is reacted with potassium hydroxide the complex salt is
potassium tetrahydroxoplumbate(II) complex salt.
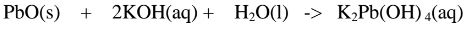
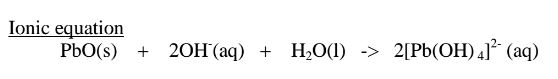
(ii)When Lead(II)hydroxide is placed in a boiling tube containing 10cm3 of either
2M nitric(V)acid or 2M sodium hydroxide hydroxide solution, it dissolves on both
the acid and the alkali/base to form a colourless solution. i.e.
(i) when reacting with nitric(V)acid, the hydroxide shows basic properties. It
reacts with the acid to form a simple salt and water only.

(ii) when reacting with sodium hydroxide, the hydroxide shows acidic properties.
It reacts with a base to form a complex salt.
Chemical equation
1.When Lead(II) hydroxide is reacted with sodium hydroxide the complex salt is
sodium tetrahydroxoplumbate(II) complex salt.

2.When Lead(II) hydroxide is reacted with potassium hydroxide the complex salt
is potassium tetrahydroxoplumbate(II) complex salt.

(c)Aluminium(III)0xide(Al2O3) and Aluminium(III)hydroxide(Al(OH)3)
(i)When 1/2 spatula full of Aluminium(III)oxide is placed in a boiling tube
containing 10cm3 of either 2M nitric(V)acid or 2M sodium hydroxide hydroxide
solution, it dissolves on both the acid and the alkali/base to form a colourless
solution. i.e.
(i) when reacting with nitric(V)acid, the oxide shows basic properties by
reacting with an acid to form a simple salt and Water only.

(ii) when reacting with sodium hydroxide, the oxide shows acidic properties by
reacting with a base to form a complex salt.
Chemical equation
1.When Aluminium(Ill) oxide is reacted with sodium hydroxide the complex salt
is sodium tetrahydroxoaluminate(III) complex salt.

2.When Aluminium(III) oxide is reacted with potassium hydroxide the complex
salt is potassium tetrahydroxoaluminate(II) complex salt.

(ii)When Aluminium(IlI)hydroxide is placed in a boiling tube containing 10cm3 of
either 2M nitric(V)acid or 2M sodium hydroxide hydroxide solution, it dissolves
on both the acid and the alkali/base to form a colourless solution. i.e.
(i) when reacting with nitric(V)acid, the hydroxide shows basic properties. It
reacts with the acid to form a simple salt and water only.

(ii) when reacting with sodium hydroxide, the hydroxide shows acidic properties.
It reacts with a base to form a complex salt.
Chemical equation
1.When aluminium(III) hydroxide is reacted with sodium hydroxide the complex
salt is sodium tetrahydroxoaluminate(III) complex salt.
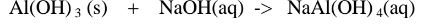
2.When aluminium(Ill) hydroxide is reacted with potassium hydroxide the
complex salt is potassium tetrahydroxoaluminate(III) complex salt.
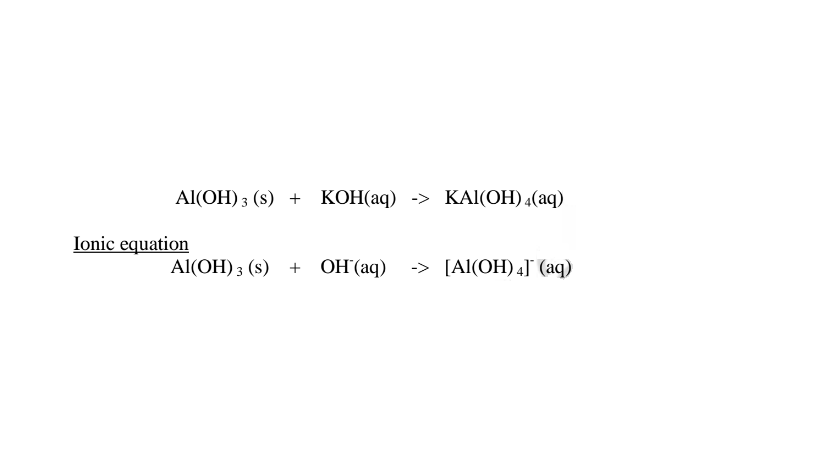
Summary of amphotellic oxides/hydroxides
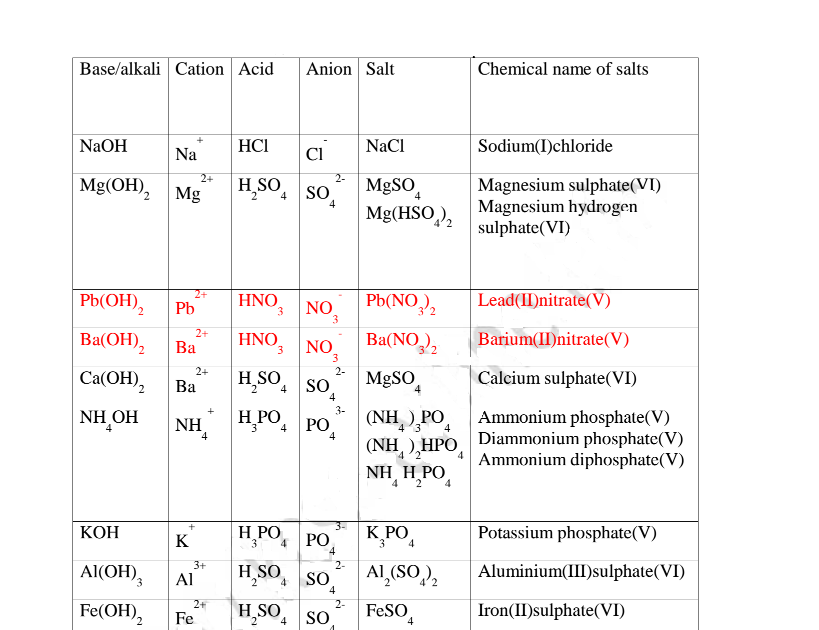
12.(a) A salt is an ionic compound formed when the cation from a base combine
with the anion derived from an acid.
A salt is therefore formed when the hydrogen ions in an acid are replaced
wholly/ fully or partially/partly ,directly or indirectly by a metal or armnonium
radical.
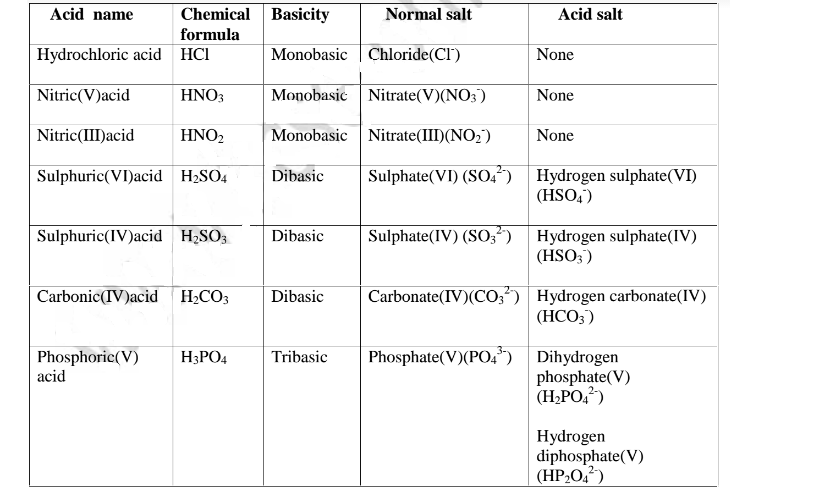
(b) The number of ionizable/replaceable hydrogen in an acid is called basicity
of an acid.
Some acids are therefore:

(c) Some salts are normal salts while other are acid salts.
(i)A normal salt is formed when all the ionizable /replaceable hydrogen in an acid is replaced by a metal or metallic /ammonium radical
(ii)An acid salt is fonned when part/portion the ionizable /replaceable hydrogen in an acid is replaced by a metal or metallic /ammonium radical.
Table showing normal and acid salts derived from common acids
The table below show shows some examples of salts.
(d) Some salts undergo hygroscopy, deliquescence and efflorescence.
(i) Hygroscopic salts /compounds are those that absorb water from the atmosphere
but do not form a solution.
Some salts which are hygroscopic include anhydrous copper(lI)sulphate(VI),
anhydrous cobalt(Il)chloride, potassium nitrate(V) common table salt.
(ii)Deliquescent salts /compounds are those that absorb water from the atmosphere
and form a solution.
Some salts which are deliquescent include: Sodium nitrate(V),Calcium chloride,
Sodium hydroxide, Iron(II)chloride, Magnesium chloride.
(iii)Efflorescent salts/compounds are those that lose their water of crystallization
to the atmosphere.
Some salts which efflorescence include: sodium carbonate decahydrate,
Iron(II)sulphate(VI)heptahydrate, sodium sulphate (VI)decahydrate.
(e)Some salts contain water of crystallization.They are hydrated.Others do not
contain water of crystallization. They are anhydrous.
Table showing some hydrated salts.

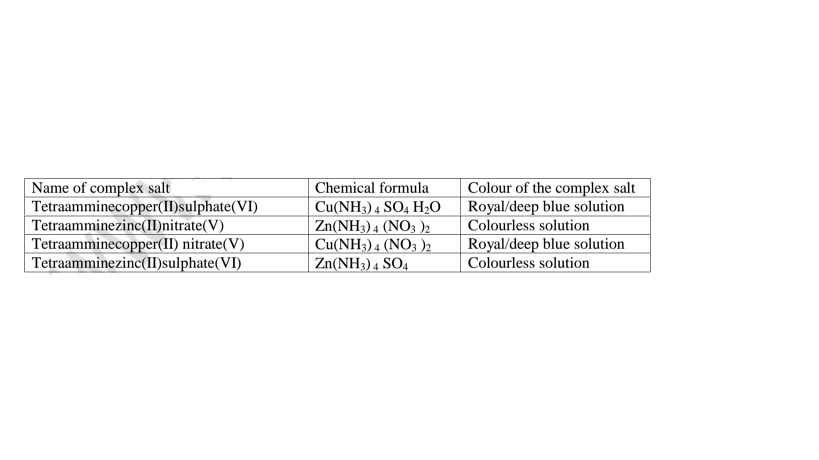
(i)Some salts exist as a simple salt while some as complex salts. Below are some
complex salts.
Table of some complex salts
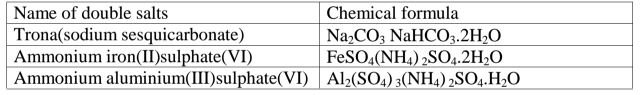
(g)Some salts exist as two salts in one. They are called double salts.
Table of some double salts
(h)Some salts dissolve in water to form a solution. They are said to be soluble.
Others do not dissolve in water. They form a suspension/precipitate in water.
Table of solubility of salts

13 Salts can be prepared in a school laboratory by a method that uses its solubility
in water.
(a) Soluble salts may be prepared by using any of the following methods:
(i)Direct displacement/reaction of a metal with an acid.
By reacting a metal higher in the reactivity series than hydrogen with a dilute
acid,a salt is formed and hydrogen gas is evolved.
Excess of the metal must be used to ensure all the acid has reacted.
When effervescence/bubbling /fizzing has stopped ,excess metal is filtered.
The filtrate is heated to concentrate then allowed to crystallize.
Washing with distilled water then drying between filter papers produces a sample
crystal of the salt. i.e.
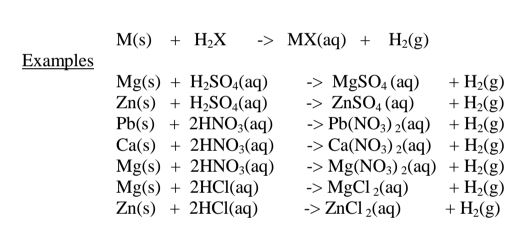
(ii)Reaction of an insoluble base with an acid
By adding an insoluble base (oxide/hydroxide )to a dilute acid until no more
dissolves, in the acid,a salt and water are formed. Excess of the base is filtered off.
The filtrate is heated to concentrate ,allowed to crystallize then washed with
distilled water before drying between filter papers e.g.

(iii)reaction of insoluble /soluble carbonate /hydrogen carbonate with an acid.
By adding an excess of a soluble /insoluble carbonate or hydrogen carbonate to
a dilute acid, effervescence /fizzing/bubbling out of carbon(lV)oxide gas shows the
reaction is taking place.
When effervescence /fizzing/bubbling out of the gas is
over, excess of the insoluble carbonate is filtered off. The filtrate is heated to
concentrate ,allowed to crystallize then washed with distilled water before drying
between filter paper papers e. g.
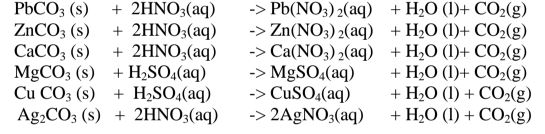
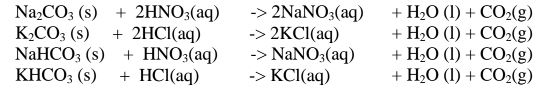
(iv)neutralization/reaction of soluble base/alkali with dilute acid
By adding an acid to a burette into a known volume of an alkali with 2-3 drops of
an indicator, the colour of the indicator changes when the acid has completely
reacted with an alkali at the end point.
The procedure is then repeated without the
indicator .The solution mixture is then heated to concentrate , allowed to crystallize
,washed with distilled water before drying with filter papers. e. g.

(iv)direct synthesis/combination.
When a metal burn in a gas jar containing a non metal , the two directly combine
to form a salt. e.g.

Some salts once formed undergo sublimation and hydrolysis. Care should be
taken to avoid water/moisture into the reaction flask during their preparation.Such
salts include aluminium(III)chloride(AlC13) and iron (III)chloride(FeCl3)
1. Heated aluminium foil reacts with chlorine to form aluminium(lII)chloride that
sublimes away from the source of heating then deposited as solid again
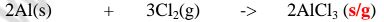
Once formed aluminium(11l)chloride hydrolyses/reacts with water vapour/
moisture present to form aluminium hydroxide solution and highly acidic fumes of
hydrogen chloride gas.
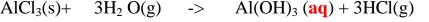
2. Heated iron filings reacts with chlorine to form iron(III)chloride that sublimes
away from the source of heating then deposited as solid again
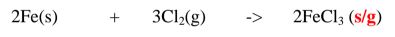
Once formed , aluminium(III)chloride hydrolyses/reacts with Water vapour/
moisture present to form aluminium hydroxide solution and highly acidic fumes of
hydrogen chloride gas.
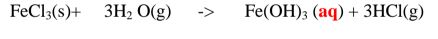
(b)Insoluble salts can be prepared by reacting two suitable soluble salts to form
one soluble and one insoluble. This is called double decomposition or
precipitation. The mixture is filtered and the residue is washed with distilled
Water then dried.

14. Salts may lose their water of crystallization , decompose ,melt or sublime on
heating on a Bunsen burner flame.
The following shows the behavior of some salts on heating gently /or strongly in a
laboratory school burner:
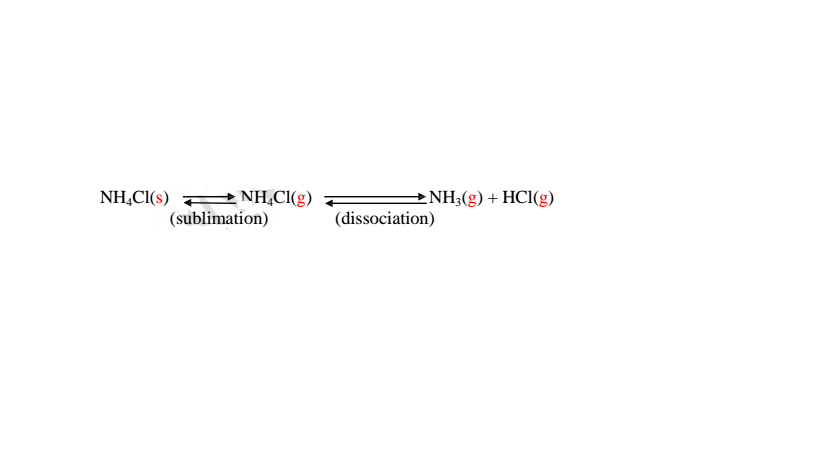
(a)effect of heat on chlorides
All chlorides have very high melting and boiling points and therefore are not
affected by laboratory heating except ammonium chloride. Ammonium chloride
sublimes on gentle heating. It dissociate into the constituent ammonia and
hydrogen chloride gases on strong heating.
(b)effect of heat on nitrate(V)
(i) Potassium nitrate(V)/KNO3 and sodium nitrate(V)/NaNO3 decompose on
heating to form Potassium nitrate(III)/KNO2 and sodium nitrate(III)/NaNO2 and
producing Oxygen gas in each case.
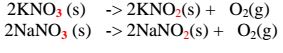
(ii)Heavy metal nitrates(V) salts decompose on heating to form the oxide and a
mixture of brown acidic nitrogen(IV)oxide and oxygen gases. e.g.
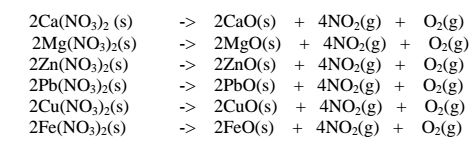
(iii)Silver(I)nitrate(V) and mercury(II) nitrate(V) are lowest in the reactivity series
They decompose on heating to form the metal(silver and mercury)and the
Nitrogen(IV)oxide and oxygen gas. i.e.
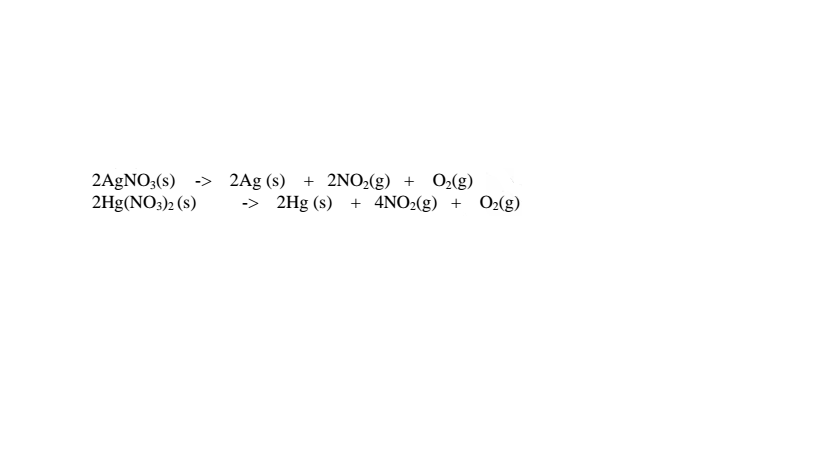
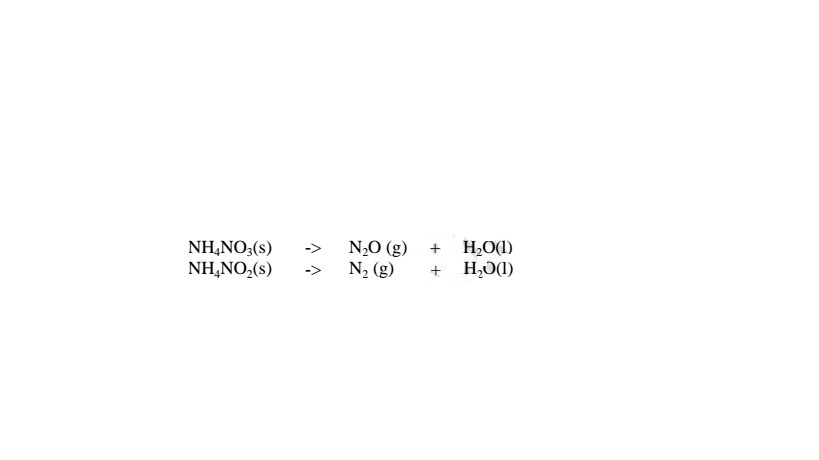
(iv)Ammonium nitrate(V) and Ammonium nitrate(III) decompose on heating to
Nitrogen(I)oxide(relights/rekindles glowing splint) and nitrogen gas
respectively.Water is also formed.i.e.
(c) effect of heat on nitrate(V)
Only Iron(II)sulphate(VI), Iron(III)sulphate(VI) and copper(II)sulphate(VI)
decompose on heating. They form the oxide, and produce highly acidic fumes of
acidic sulphur(IV)oxide gas.

(d) effect of heat on carbonates(IV) and hydrogen carbonate(IV).
(i)Sodium carbonate(IV)and potassium carbonate(IV)do not decompose on
heating.
(ii)Heavy metal nitrate(IV)salts decompose on heating to form the oxide and
produce carbon(IV)oxide gas. Carbon (IV)oxide gas forms a white precipitate
when bubbled in lime Water. The White precipitate dissolves if the gas is in excess

(iii)Sodium hydrogen carbonate(IV) and Potassium hydrogen
carbonate(IV)decompose on heating to give the corresponding carbonate (IV) and
form water and carbon(IV)oxide gas. i.e.
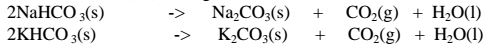
(iii) Calcium hydrogen carbonate (IV) and Magnesium hydrogen carbonate(IV)
decompose on heating to give the corresponding carbonate (IV) and form water
and carbon(IV)oxide gas. i. e.
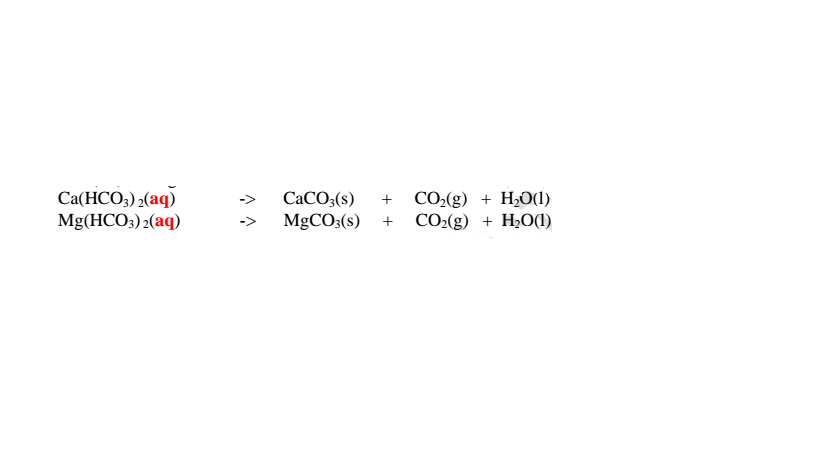
15. Salts contain cation(positively charged ion) and anions(negatively charged
ion).When dissolved in polar solvents/water.
The cation and anion in a salt is determined/known usually by precipitation of the
salt using a precipitating reagent.
The colour of the precipitate is a basis of qualitative analysis of a compound.
16.Qualitative analysis is the process of identifying an unknown compound /salt by
identifying the unique qualities of the salt/compound.
It involves some of the following processes.
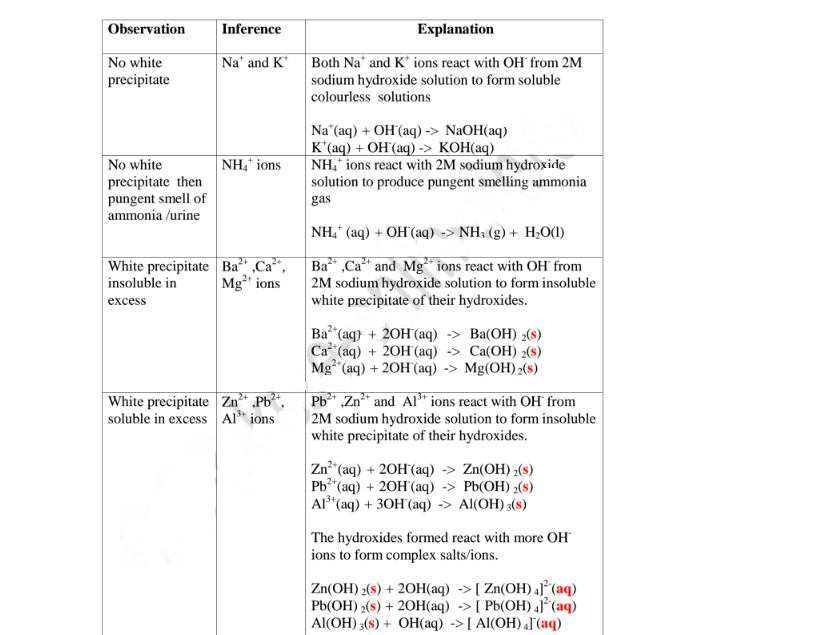
(a)Reaction of cation with sodium/potassium hydroxide solution.
Both sodium/potassium hydroxide solutions are precipitating reagents.
The alkalis produce unique colour of a precipitate/suspension when a few/three
drops is added and then excess alkali is added to unknown salt/compound
solution.
NB: Potassium hydroxide is not commonly used because it is more expensive than
sodium hydroxide.
The table below shows the observations, inferences / deductions and explanations
from the following test tube experiments:
Procedure
Put about 2cm3 of MgCl2, CaCl2, AlCl3, NaCl, KCI, FeSO4, Fe2(SO4)3, CuSO4,
ZnSO4NH4NO3, Pb(NO3)2, Ba(NO3)2 each into separate test tubes. Add three
drops of 2M sodium hydroxide solution then excess (2/3 the length of a standard
test tube).

(b)Reaction of cation with aqueous ammonia
Aqueous ammonia precipitating reagent that can be used to identify the cations
present in a salt.
Like NaOH/KOH the OH’ ion in NH4OH react with the cation to form a
characteristic hydroxide .
Below are the observations ,inferences and explanations of the reactions of
aqueous ammonia with salts from the following test tube reactions.
Procedure
Put about 2cm3 of MgCl2, CaCl2, AlCl3, NaCl, KCI, FeSO4, Fe2(SO4)3, CuSO4,
ZnSO4NH4NO3, Pb(NO3)2, Ba(NO3)2 each into separate test tubes.
Add three drops of 2M aqueous ammonia then excess (2/3 the length of a standard
test tube).
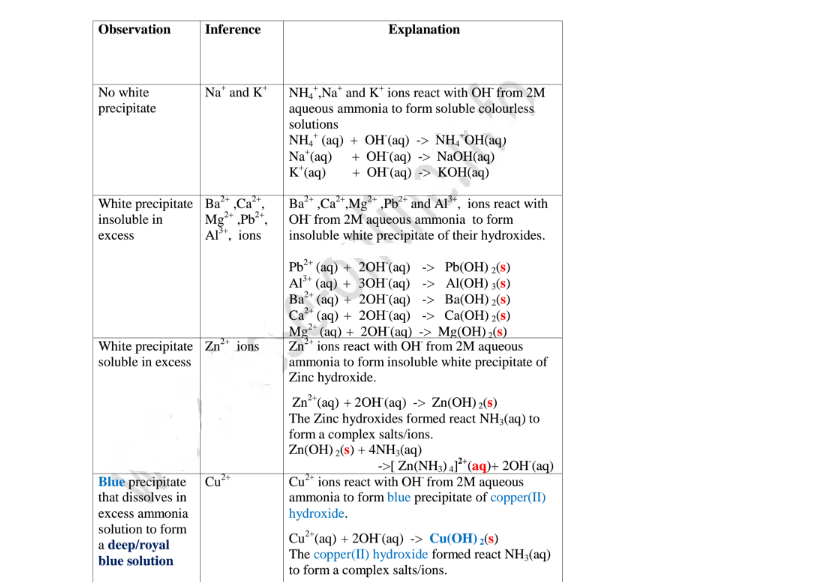

Note
(i) Only Zn2+ ions/salts form a white precipitate that dissolve in excess of both
2M sodium hydroxide and 2M aqueous ammonia.
(ii) Pb2+ and Al3+ ions/salts form a white precipitate that dissolve in excess of 2M
sodium hydroxide but not in 2M aqueous ammonia.
(iii) Cu2+ ions/salts form a blue precipitate that dissolve to form a deep/royal
blue solution in excess of 2M aqueous ammonia but only blue insoluble
precipitate in 2M sodium hydroxide
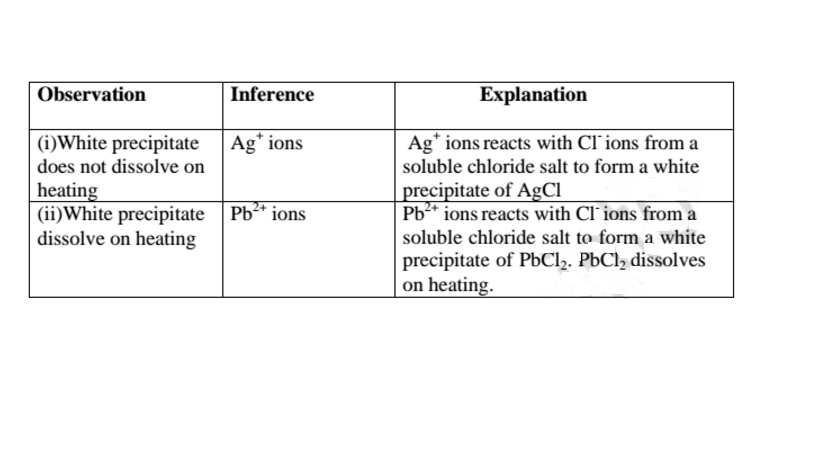
(c)Reaction of cation with Chloride (Cl-)ions
All chlorides are soluble in water except Silver chloride and Lead (II)chloride
(That dissolve in hot Water).When a soluble chloride like NaCl, KCI, NH4Cl is
added to about 2cm3 of a salt containing Ag+ or Pb2+ ions a White precipitate of
AgCl or PbCl2 is formed. The following test tube reactions illustrate the above.
Experiment
Put about 2cm3 of silver nitrate(V) and Lead(II)nitrate(V)solution into separate test
tubes. Add five drops of NaCl /KCl / NH4Cl/HCl. Heat to boil.
Note
Both Pb2+ and Al3+ ions forms an insoluble white precipitate in excess aqueous
ammonia. A white precipitate on adding Cl’ ions/salts shows Pb2+.
No white precipitate on adding Cl‘ ions/salts shows Al3+.
Adding a chloride/ Cl’ ions/salts can thus be used to separate the identity of Al3+
and Pb2+.
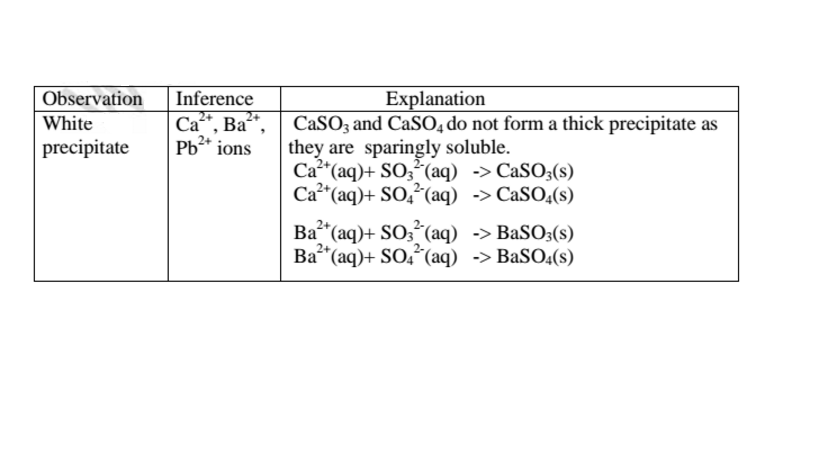
(d)Reaction of cation with sulphate(VI)/SO42‘ and sulphate(IV)/S03} ions
All sulphate(VI) and sulphate(IV)/SO32″ ions/salts are soluble/dissolve in water
except Calcium sulphate(VI)/CaSO4, Calcium sulphate(IV)/CaSO3, Barium
sulphate(VI)/BaSO4, Barium sulphate(IV)/BaSO3, Lead(II) sulphate(VI)/PbSO4
and Lead(II) sulphate(IV)/PbSO3.When a soluble sulphate(VI)/SO42′ salt like
Na2SO4, HZSO4, (NH4)2SO4 or Na2SO3 is added to a salt containing Ca2+, Pb”,
Ba” ions, a white precipitate is formed.
The following test tube experiments illustrate the above.
Procedure
Place about 2cm3 of Ca(NO3)2, Ba(NO3)2, BaCl2 and Pb(NO3)2, in separate
boiling tubes. Add six drops of sulphuric(VI)acid /sodium sulphate(VI)/ammonium
sulphate(VI)solution. Repeat with six drops of sodium sulphate(IV).
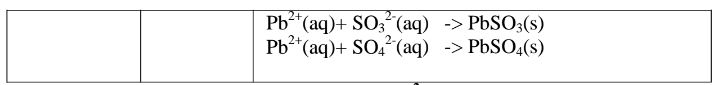
(e)Reaction of cation with carbonate(IV)/C03 2- ions
All carbonate salts are insoluble except sodium/potassium carbonate(IV) and
ammonium carbonate(IV).
They dissociate /ionize to release CO3 2- ions. CO3 2- ions produce a white
precipitate when the soluble carbonate salts is added to any metallic cation.
Procedure
Place about 2cm3 of Ca(NO3)2, Ba(NO3)2, MgCl2 ,Pb(NO3)2 and ZnSO4 in separate
boiling tubes.
Add six drops of Potassium /sodium carbonate(IV)/ ammonium carbonate
(IV)solution.
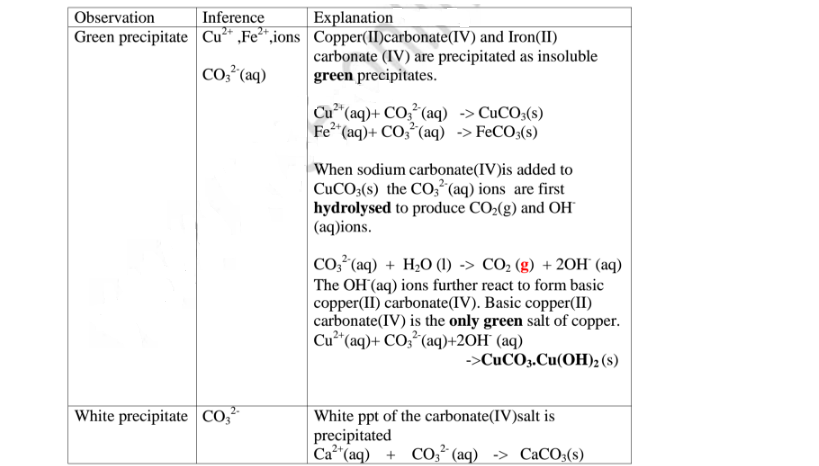
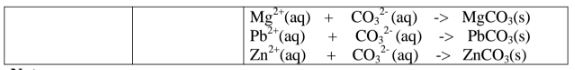
Note
(i)Iron(III)carbonate(IV) does not exist.
(ii)Copper(ll)Carbonate(IV) exist only as the basic CuCO3.Cu(OH) 2
(iii)Both BaCO3 and BaSO; are insoluble White precipitate. If hydrochloric
acid is added to the white precipitate;
I. BaCO3 produces CO2 gas. When bubbled/directed into lime water
solution,a white precipitate is formed.
II. I. BaSO3 produces SO; gas. When bubbled/directed into orange
acidified potassium dichromate(VI) solution, it turns to
green/decolorizes acidified potassium manganate(VII).
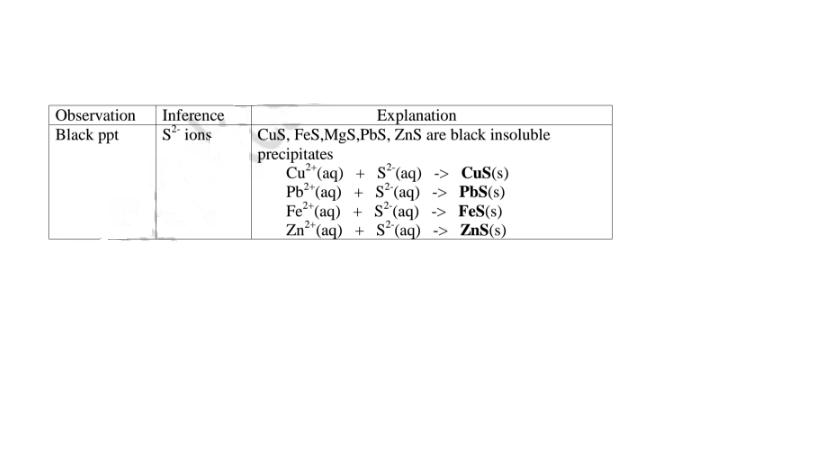
(f) Reaction of cation with sulphide / S2- ions
All sulphides are insoluble black solids/precipitates except sodium sulphide/ Na2S/
potassium sulphide/K2S. When a few/3 drops of the soluble sulphide is added to a
metal cation/salt, a black precipitate is formed.
Procedure
Place about 2cm3 of Cu(NO3)2, F eSO4, MgCl2,Pb(NO3)2 and ZnSO4 in separate
boiling tubes.
Add six drops of Potassium /sodium sulphide solution.
Sample qualitative analysis guide
You are provided with solid Y(aluminium (III)sulphate(VI)hexahydrate).Carry out
the following tests and record your observations and inferences in the space
provided.

(b)Place about a half spatula full of the solid into a clean dry boiling tube. Heat
gently then strongly.

(c)Place all the remaining portion of the solid in a test tube .Add about 10cm3 of
distilled water. Shake thoroughly. Divide the mixture into five portions.

(i)To the first portion, add three drops of sodium hydroxide then add excess of the
alkali.

(ii)To the second portion, add three drops of aqueous ammonia then add excess of
the alkali.

(iii)To the third portion, add three drops of sodium sulphate(VI)solution.

(iv)I.To the fourth portion, add three drops of Lead(II)nitrate(IV)solution.
Preserve

II.To the portion in (iv) I above , add five drops of dilute hydrochloric acid.

III.To the portion in (iv) ll above, heat to boil.

Note that:
(i)From test above, it can be deduced that solid Y is hydrated
aluminium(lII)sulphate(VI) solid
(ii)Any ion inferred from an observation below must be derived from previous
correct observation and inferences above. e.g.
Al“ in c(m) must be correctly inferred in either/or in c(ii) or c(i)above
SO42′ in c(iv)III must be correctly inferred in either/or in c(iv)lI or c(iv)
above
(iii)Contradiction in observations and inferences should be avoided eg.
“White ppt soluble in excess” to infer presence of Al“ ,Q“ ,Pb3+
(iv)Symbols of elements/ions should be correctly capitalized. e.g.
“SO4’2” is Wrong, “sO42″’ is Wrong, “cuzl” is Wrong.
Sample solutions of salt were labeled as I,II, III and IV. The actual solutions,
not in that order are lead nitrate, zinc sulphate potassium chloride and
calcium chloride.
a)When aqueous sodium carbonate was added to each sample separately, a
white precipitate was formed in I, III and IV only. Identify solution II.
b)When excess sodium hydroxide was added to each sample separately, a
white precipitate was formed in solutions III and I only.
Identify solution I
17.When solids/salts /solutes are added to a solvent ,some dissolve to form a
solution.
Solute + Solvent -> Solvent
If a solution has a lot of solute dissolved in a solvent ,it is said to be concentrated.
If a solution has little solute dissolved in a solvent ,it is said to be dilute.
There is a limit to how much solute can dissolve in a given /specified amount of
solvent/water at a given /specified temperature.
The maximum mass of salt/solid/solute that dissolve in 100g of solvent/water
at a specified temperature is called solubility of a salt.
When no more solute can dissolve in a given amount of solvent at a specified
temperature, a saturated solution is formed.
For some salts, on heating, more of the salt/solid/solute dissolve in the saturated
solution to form a super saturated solution.
The solubility of a salt is thus calculated from the formula
Solubility = Mass of solute/salt/solid x 100 divided by Mass/volume of water/solvent
Practice examples
(a)Calculate the solubility of potassium nitrate(V) if 5.0 g of the salt is
dissolved in 50.0cm3 of water.
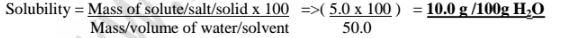
(b)Calculate the solubility of potassium chlorate(V) if 50.0 g of the salt is
dissolved in 250.0cm3 of water.
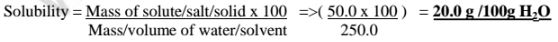
(c)If the solubility of potassium chlorate(V) is 5g/100g H10 at 80°C,how much
can dissolve in 5cm3 of Water at 80°C .
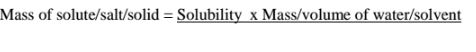
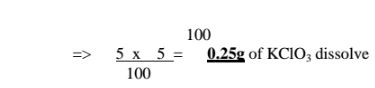
(d)If the solubility of potassium chlorate(V) is 72g/100g H20 at 20°C,how
much can saturate 25g of Water at 20°C .
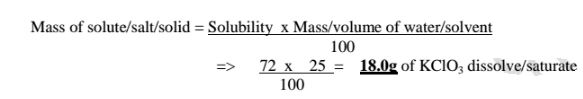
(e) 22g of potassium nitrate(V) was dissolved in 40.0g of Water at 10°C.
Calculate the solubility of potassium nitrate(V) at 10°C.
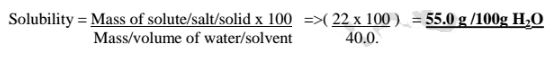
(f)What volume of water should be added to 22.0g of water at 10°C if the
solubility of KNO3 at 10°C is 5.0g/100g H20?
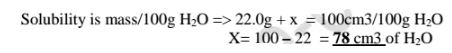
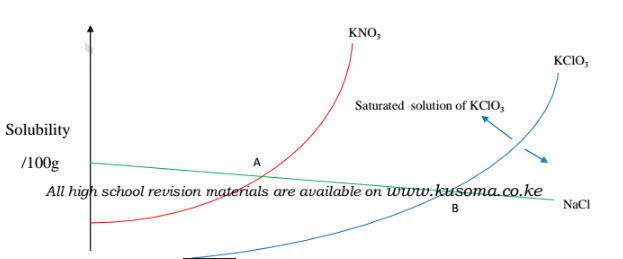
18. A graph of solubility against temperature is called solubility curve.
It shows the influence of temperature on solubility of different
substances/solids/salts.
Some substances dissolve more with increase in temperature while for others
dissolve less with increase in temperature
Note:
(i)solubility of KNO3 and KClO3 increase with increase in temperature.
(ii)solubility of KNO3 is always higher than that of KCIO3 at any specified
temperature.
(iii)solubility of NaCl decrease with increase in temperature.
(iv)NaCl has the highest solubility at low temperature while KCIO3 has the lowest
solubility at low temperature.
(v)At point A both NaCl and KNO3 are equally soluble.
(vi)At point B both NaCl and KCIO3 are equally soluble.
(vii) An area above the solubility curve of the salt shows a saturated
/supersaturated solution.
(viii) An area below the solubility curve of the salt shows an unsaturated
solution.
19.(a) For salts whose solubility increases with increase in temperature, crystals
form when the salt solution at higher temperatures is cooled to a lower
temperature.
(b) For salts whose solubility decreases with increase in temperature, crystals
form when the salt solution at lower temperatures is heated to a higher
temperature.
The examples below shows determination of the mass of crystals deposited with
changes in temperature.
1.The solubility of KCIO3 at 100°C is 60g/100g water .What mass of KCIO3
will be deposited at:
(i)75 “C if the solubility is now 39g/100g water.
At 100°C = 60.0g
Less at 75°C = – 39.0g
Mass of crystallized out 21.0 g
(i)35 “C if the solubility is now 28 g/ 100g water.
At 100°C = 60.0g
Less at 35°C = – 28.0.0g
Mass of crystallized out = 32.0g
2. KNO3 has a solubility of 42 g/100g water at 20°C.The salt was heated and
added 38g more of the solute which dissolved at 100°C. Calculate the solubility
of KNO3 at 100°C.
Solubility of KNO3 at 100°C = solubility at 20°C + mass of KNO3 added
=> 42g + 38g = so KNO3/100 HZQ
3. A salt solution has a mass of 65g containing 5g of solute. The solubility of
this salt is 25g per 100g water at 20°C. 60g of the salt are added to the solution
at 20°C.Calculate the mass of the solute that remain undissolved.

4. Study the table below
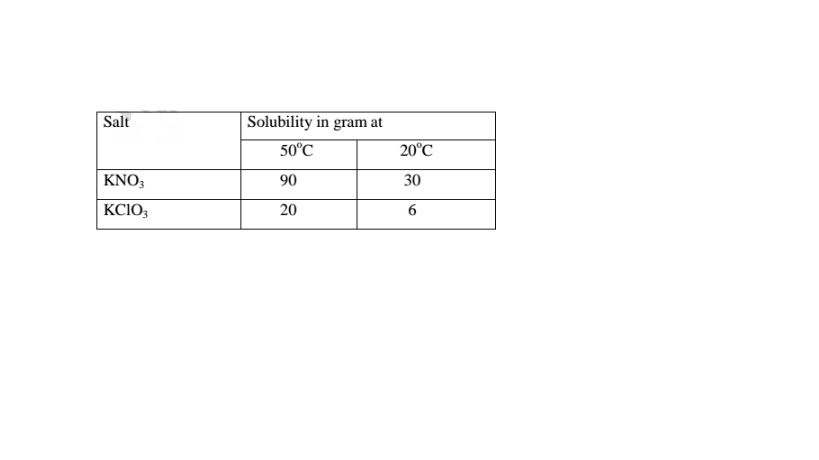
(i)What happens when the two salts are dissolved in water then cooled from
50°C to 20°C.
(90 — 30) = 60.0 g of KN03 crystals precipitate
(20 — 6) = 14.0 g of KC103 crystals precipitate
(ii)State the assumption made in (i) above.
Solubility of one salt has no effect on the solubility of the other.
5. 10.0 g of hydrated potassium carbonate (IV) K2CO3.XH20 on heating leave
7.93 of the hydrate.
(a)Calculate the mass of anhydrous salt obtained.
Hydrated on heating leave anhydrous = 7.93 g
(b)Calculate the mass of water of crystallization in the hydrated salt
Mass of Water of crystallization = hydrated – anhydrous
-> 10.0 – 7.93 = 2.07 g
(c)How many moles of anhydrous salt are there in 10 of hydrate? (K=
39.0,C=12.0.0= 16.0)

(d)How many moles of water are present in the hydrate for every one mole of
K2C03 ? (H=1.0.0= 16.0)

(e)What is the formula of the hydrated salt?
K2C03 .2H20
6. The table below shows the solubility of Potassium nitrate(V) at different
temperatures.


(a)Plot a graph of mass of in 100g water(y-axis) against temperature in °C
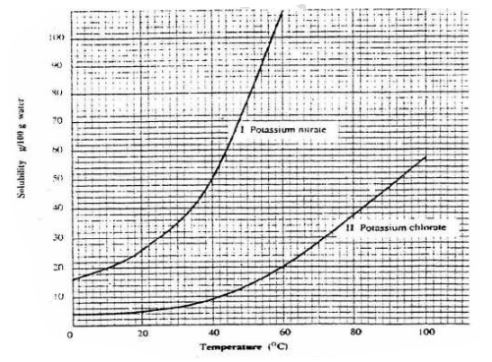
(b)From the graph show and determine
(i)the mass of KNO3 dissolved at:
l.1. 20°C
From a correctly plotted graph = 32g
II. 35°C
From a correctly plotted graph = 57g
III. 55°C
From a correctly plotted graph = 104g
(ii)the temperature at which the following mass of KNO3 dissolved:
I. 22g
From a correctly plotted graph Il3.0°C
II. 30g
From a correctly plotted graph Il7.5°C
III.100g
From a correctly plotted graph I54.5°C
(c)Explain the shape of your graph.
Solubility of KNO3 increase with increase in temperature/More KNO3 dissolve as
temperature rises.
(d)Show on the graph the supersaturated and unsaturated solutions.
Above the solubility curve Write; “supersaturated”
Below the solubility curve write; “unsaturated”
(e)From your graph, calculate the amount of crystals obtained when a
saturated solution of KNO3 containing 180 g of the salt is cooled from 80°C to
I. 20°C
Solubility before heating = 180 g
Less Solubility after heating(from the graph) = 32 g
Mass of KNO3 crystals = 148 g
II. 35°C
Solubility before heating = 180 g
Less Solubility after heating(from the graph) = 58 g
Mass of KNO3 crystals = 122 g
III. 55°C
Solubility before heating = 180 g
Less Solubility after heating(from the graph) 102 g
Mass of KNO3 crystals = 78 g
7. The table below shows the solubility of salts A and B at various
temperatures.
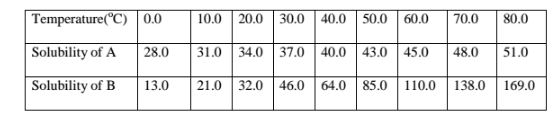
(a)On the same axis plot a graph of solubility (y-axis) against temperature for
each salt.
(b)At what temperature are the two salts equally soluble.
The point of intersection of the two curves = 24°C
(c)What happens when a mixture of 100 g of salt B with 100 g if water is heated
to 80°C
From the graph, the solubility of B at 80°C is 169 g /100 g water. All the 100 g
crystals of B dissolve.
(d)What happens when the mixture in (c) above is then cooled from 50°C to
20°C.
Method I.
Total mass before cooling at 50°C = 100.0 g
(From graph) Solubility/mass after cooling at 20°C = 32.0 g
Mass of crystals deposited = 68.0 g
Method II.
Mass of soluble salt crystals at 50°C added = 100 g
(From graph)Solubility/mass before cooling at 50°C = 85.0 g
Mass of crystals that cannot dissolve at 50°C = 15.0 g
(From graph) Solubility/mass before cooling at 50°C = 85.0 g
(From graph) Solubility/mass after cooling at 20°C = 32.0 g
Mass of crystals deposited after cooling = 53.0 g
Total mass of crystals deposited = 15.0 + 53.0 = 68.0 g
(e)A mixture of 40 g of A and 60 g of B is added to 10 g of water and heated to
70°C.The solution is then allowed to cool to 10°C.Describe clearly what
happens.
I.For salt A
Solubility of A before heating = mass of A x 100 divide by Volume of water added
=> 40 x 100 divide by 10
=400g/100g Water
(Theoretical)Solubility of A before heating = 400 g
Less (From graph ) Solubility of A after heating at 70°C = 48 g
Mass of crystals that can not dissolve at 70°C = 352 g
(From graph ) Solubility of A after heating at 70°C = 48 g
Less (From graph) Solubility of A after cooling to 10°C = 31 g
Mass of crystals that crystallize out on cooling to 10°C = 17 g
Mass of crystals that can not dissolve at 70°C = 352 g
Add Mass of crystals that crystallize out on cooling to 10°C = 17 g
Total mass of A that does not dissolve/crystallize/precipitate = 369 g
I.For salt B
Solubility of B before heating = mass of B X 100 divide by Volume of water added
=> 60 x 100 divide by 10
= 600g/100g Water
(Theoretical)Solubility of B before heating = 600 g
Less (From graph ) Solubility of B after heating at 70°C = 138 g
Mass of crystals that cannot dissolve at 70°C = 462 g
(From graph ) Solubility of B after heating at 70°C = 138 g
Less (From graph) Solubility of B after cooling to 10°C = 21 g
Mass of crystals that crystallize out on cooling to 10°C = 117 g
Mass of crystals that cannot dissolve at 70°C = 462 g
Add Mass of crystals that crystallize out on cooling to 10°C = 117 g
Total mass of A that does not dissolve/crystallize/precipitate = 579 g
(f)State the assumption made in (e)above
Solubility of one salt has no effect on the solubility of the other
8. When 5.0 g of potassium chlorate (V) was put in 10cm3 of water and
heated, the solid dissolves. When the solution was cooled , the temperature at
which crystals reappear was noted. Another 10cm3 of Water was added and
the mixture heated to dissolve then cooled for the crystals to reappear .The
table below shows the the results obtained
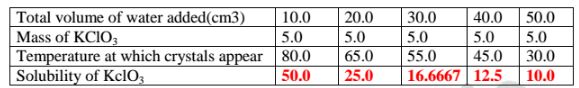
(a)Complete the table to show the solubility of KclO3 at different
temperatures.
(b)Plot a graph of mass of KCIO3 per 100g water against temperature at
which crystals form.
(c)From the graph, show and determine ;
(i)the solubility of KCIO3 at
I. 50°C
From a well plotted graph = 14.5 g KCIO3/100g Water
II. 35°C
From a well plotted graph = 9.0 g K0103/100g water
(ii)the temperature at which the solubility is:
I. 10g/100g water
From a Well plotted graph = 38.0 °C
lI. 45g/100g water
From a well plotted graph = 77.5 °C
(d)Explain the shape of the graph.
Solubility of KCIO3 increase with increase in temperature/more KclO;dissolve as
temperature rises.
(e)What happens when 100g per 100g water is cooled to 35.0 °C
Solubility before heating = 100.0
(From the graph) Solubility after cooling = 9.0
Mass of salt precipitated/crystallization = 91.0 g
9. 25.0cm3 of water dissolved various masses of ammonium chloride crystals
at different temperatures as shown in the table below.
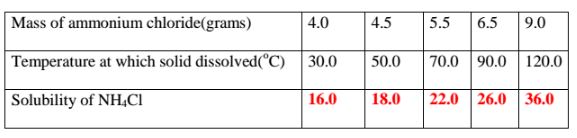
(a)Complete the table
(b)Plot a solubility curve
(c)What happens when a saturated solution of ammonium chloride is cooled
from 80°C to 40°C.
(From the graph )Solubility at 80°C = 24.0 g
Less (From the graph )Solubility at 40°C = 16.8 g
Mass of crystallized/precipitated = 7.2 g
20. Solubility and solubility curves are therefore Q
(i) to know the effect of temperature on the solubility of a salt
(ii)to fractional crystallize two soluble salts by applying their differences in
solubility at different temperatures.
(iii)determine the mass of crystal that is obtained from crystallization.
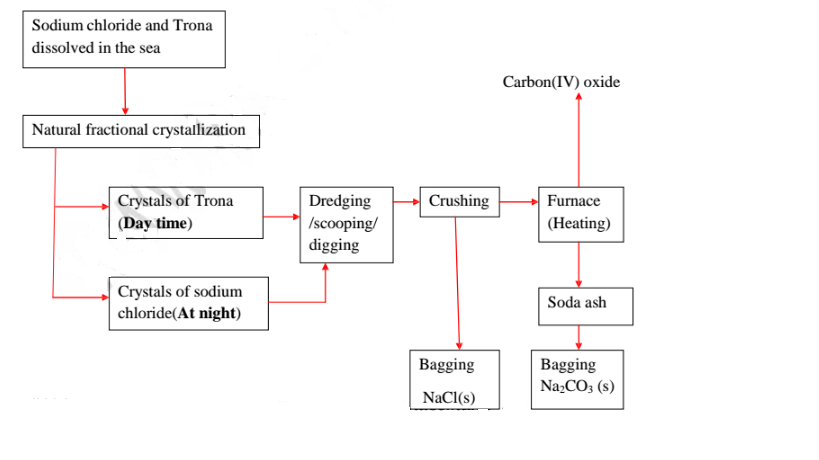
21.Natural fractional crystallization takes place in Kenya/East Africa at:
(i) Lake Magadi during extraction of soda ash(Sodium carbonate) from
Trona(sodium sesquicarbonate)
(ii) Ngomeni near Malindi at the Indian Ocean Coastline during the
extraction of common salt(sodium chloride).
22.Extraction of soda ash from Lake Magadi in Kenya
Rain water drains underground in the great rift valley and percolate underground
where it is heated geothermically.
The hot water dissolves underground soluble sodium compounds and comes out on
the surface as alkaline springs which are found around the edges of Lake Magadi
in Kenya.
Temperatures around the lake are very high (30-40°C) during the day.
The solubility of trona decrease with increase in temperature therefore solid
crystals of trona grows on top of the lake (up to or more than 30 metres thick)
A bucket dredger mines the trona which is then crushed ,mixed with lake liquor
and pumped to washery plant where it is further refined to a green granular product
called CRS.
The CRS is then heated to chemically decompose trona to soda ash(Sodium
carbonate)
Soda ash(Sodium carbonate) is then stored .It is called Magadi Soda. Magadi Soda
is used :
(i) make glass
(ii) for making soapless detergents
(iii) softening hard Water.
(iv)
Common salt is collected at night because its solubility decreases with decrease in
temperature. It is used as salt lick/feed for animals.
Summary flow diagram showing the extraction of Soda ash from Trona
23.Extraction of common salt from Indian Ocean at Ngomeni in Kenya
Oceans are salty.They contain a variety of dissolved salts (about 77% being
sodium chloride).
During high tide ,water is collected into shallow pods and allowed to crystallize as
evaporation takes place.The pods are constructed in series to increase the rate of
evaporation.
At the final pod ,the crystals are scapped together,piled in a heap and washed with
brine (concentrated sodium chloride).
It contains MgCl2 and CaCl2 . MgCl2 and CaCl2 are hygroscopic. They absorb
water from the atmosphere and form a solution.
This makes table salt damp/wet on exposure to the atmosphere.
24.Some water form lather easily with soap while others do not.
Water which form lather easily with soap is said to be “soft”
Water which do not form lather easily with soap is said to be “hard”
Hardness of water is caused by the presence of Ca2+ and Mg“ ions.
Ca2+ and Mg” ions react with soap to form an insoluble grey /white
suspension/precipitate called Scum/ curd. Ca2+ and Mg“ ions in water come from
the water sources passing through rocks containing soluble salts of Ca2+ and Mg2+
e. g. Limestone or gypsum
There are two types of water hardness:
(a)temporary hardness of water
(b)permanent hardness of water
(a)temporary hardness of water
Temporary hardness of water is caused by the presence of dissolved calcium
hydrogen carbonate/Ca(HCO3)2 and magnesium hydrogen carbonate/Mg(HCO3)2
When rain water dissolve carbon(IV) oxide from the air it forms waek
carbonic(lV) acid i.e.
CO2(g) + H20(l) -> H2CO3(aq)
When carbonic(lV) acid passes through limestone/dolomite rocks it reacts to form
soluble salts i.e.
In limestone areas; H2CO3(aq) + CaCO3(s) -> Ca(HCO3)2 (aq)
In dolomite areas; H2CO3(aq) + MgCO3(s) -> Mg(HCO3)2 (aq)
(b)permanent hardness of water
Permanent hardness of water is caused by the presence of dissolved calcium
sulphate(VI)/CaSO4 and magnesium sulphate(VI)/Mg S04 Permanent hardness of
water is caused by Water dissolving CaSO4 and MgSO4 from ground rocks.
Hardness of water can be removed by the following methods:
(a)Removing temporary hardness of Water
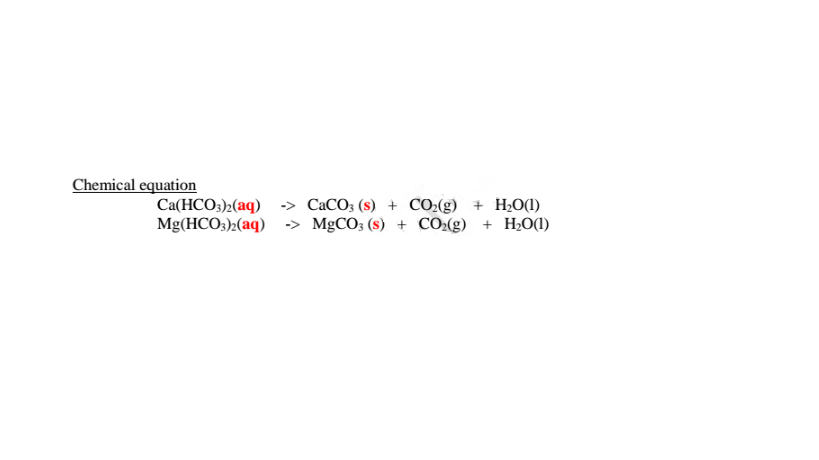
(i)Boiling/heating.
Boiling decomposes insoluble calcium hydrogen carbonate/Ca(HCO3)2 and
magnesium hydrogen carbonate/Mg(HCO3)2 to insoluble CaCO3 and MgCO3
that precipitate away. i.e
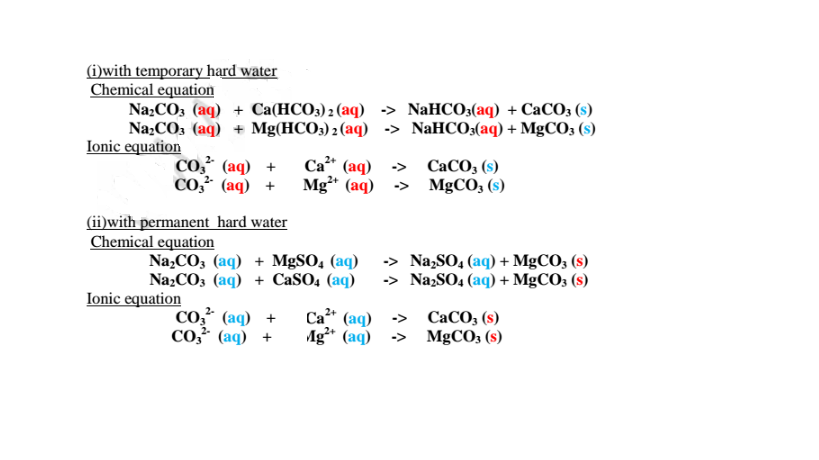
(ii)Adding sodium carbonate (IV) /Washing soda.
Since boiling is expensive on a large scale ,a calculated amount of sodium
carbonate decahydrate /Na2CO3.10H2O precipitates insoluble Ca2+(aq) and
Mg2+(aq) ions as carbonates to remove both temporary and permanent hardness of
Water .This a double decomposition reaction where two soluble salts form an
insoluble and soluble salt. i.e.
(iii)Adding calcium (Il)hydroxide/Lime water
Lime water/calcium hydroxide removes only temporary hardness of water from by
precipitating insoluble calcium carbonate(IV).
Chemical equation
Ca(OH)2 (aq) + Ca(HCO3)2(aq) -> 2H20(l) +2CaCO3 (s)
Excess of Lime water/calcium hydroxide should not be used because it dissolves
again to form soluble calcium hydrogen carbonate(lV) causing the hardness again.
(iv)Adding aqueous ammonia
Aqueous ammonia removes temporary hardness of water by precipitating
insoluble calcium carbonate(IV) and magnesium carbonate(IV)
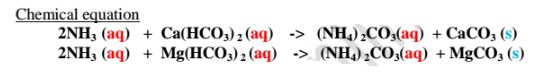
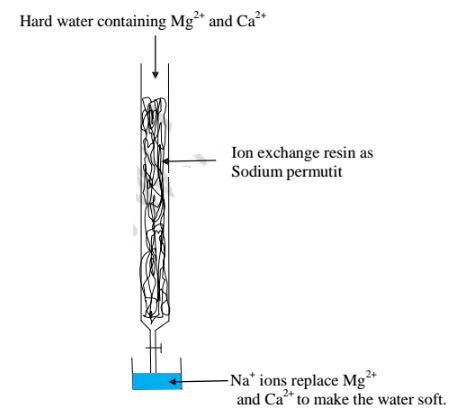
(v)Use of ion-exchange permutit
This method involves packing a chamber with a resin made of insoluble complex
of sodium salt called sodium permutit.
The sodium pennutit releases sodium ions that are exchanged with Mg2+ and Ca2+
ions in hard Water making the Water to be soft. i.e.
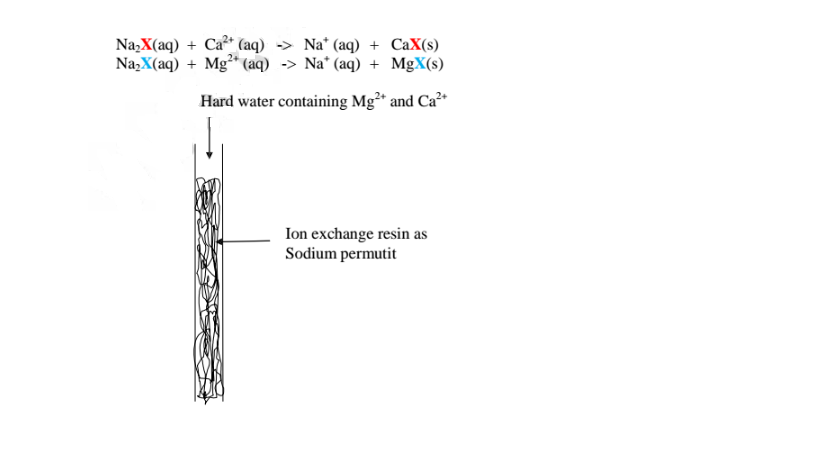
When all the Na+ ions in the resin is fully exchanged with Ca2+ and Ng2+ ions
in the permutit column ,it is said to be exhausted.
Brine /concentrated sodium chloride solution is passed through the permutit column to regenerated recharge the column again.
(vi)Deionization /demineralization
This is an advanced ion exchange method of producing deionized water .Deionized
water is extremely pure water made only of hydrogen and oxygen only without any
dissolved substances.
Deionization involve using the resins that remove all the cations by using:
(i)A cation exchanger which remove /absorb all the cations present in water and
leave only H+ ions.
(ii)An anion exchanger which remove /absorb all the anions present in water and
leave only OH‘ ions.
The H+(aq) and OH’ (aq) neutralize each other to form 1% water.
Chemical equation
H+(aq) + OH- (aq) -> H2O(l)
When exhausted the cation exchanger is regenerated by adding H+(aq) from
sulphuric(VI)acid/hydrochloric acid.
When exhausted the anion exchanger is regenerated by adding OH-(aq) from
sodium hydroxide.
Advantages of hard water
Hard water has the following advantages:
(i)Ca2+(aq) in hard water are useful in bone and teeth formation
(ii) is good for brewing beer
(iii)contains minerals that cause it to have better /sweet taste
(iv)animals like snails and coral polyps use calcium to make their shells
and coral reefs respectively.
(v)processing mineral water
Disadvantages of hard water
Hardness of water:
(i)waste a lot of soap during washing before lather is formed.
(ii)causes stains/blemishes/marks on clothes/garments
(iii)causes fur on electric appliances like kettle ,boilers and pipes form
decomposition of carbonates on heating .This reduces their efficiency hence
more/higher cost of power/electricity.
Sample revision questions
In an experiment, soap solution was added to three separate samples of water. The
table below shows the volumes of soap solution required to form lather with
1000cm3 of each sample of water before and after boiling.
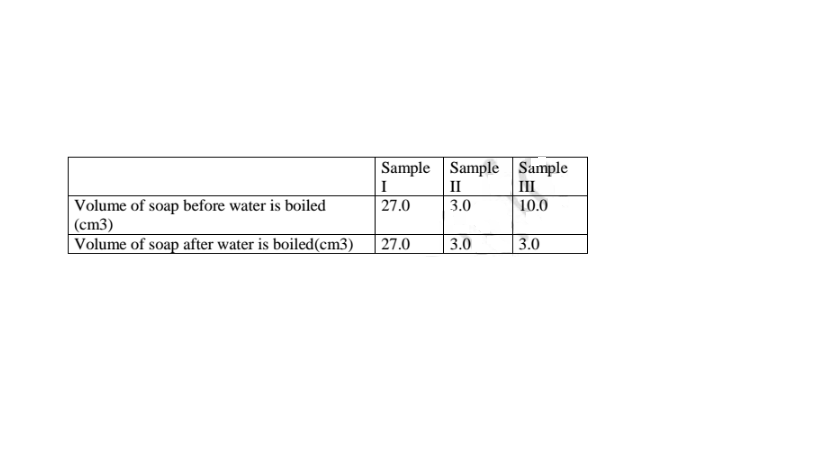
a) Which water sample is likely to be soft? Explain. (2 marks)
Sample II: Uses little sample of soap .
b) Name the change in the volume of soap solution used in sample III
(1 mark)
On heating the sample water become soft because it is temporary hard.
2.Study the scheme below and use it to answer the questions that
follow:
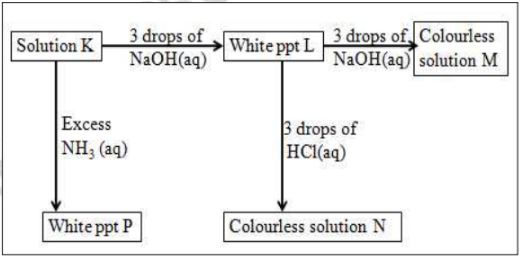
(a)Write the formula of:
(i)Cation in solution K
A13+
(ii)white ppt L
Al(OH)3
(iii) colourless solution M
[A1(OH)4]-
(iv) colourless solution N
AlCl3
(v)White ppt P
Al(OH)3
(b)Write the ionic equation for the reaction for the formation of:
(i)white ppt L
A13 +(aq) + 3OH- (aq) -> Al(OH)3(s)
(v)White ppt P
Al3+(aq) + 3OH-(aq) -> Al(OH)3(s)
(c)What property is illustrated in the formation of colourless solution M and N
Amphotellic
KCSE Revision Notes Form 1 – Form 4 All Subjects
KCSE Results » KCSE Results Top 100 Schools – Kenya Certificate of Secondary Education – KCSE » KCSE Top 100 Candidates » Kenya Certificate of Secondary Education – KCSE » KNEC – Kenya National Examinations Council » Secondary Schools in Kenya » KNEC – Kenya National Examinations Council » Free KNEC KCSE Past Papers
Kenya Scholarships for Undergraduate Students » Kenya Scholarships for Postgraduate Students » Undergraduate Scholarships for Kenyan Students » Kenya Undergraduate Scholarships » Full Undergraduate Scholarships for Kenyans » Kenya Postgraduate Scholarships » Scholarships & Grants » Undergraduate Scholarships » Universities in Kenya » Kenya Universities and Colleges Central Placement Service (KUCCPS) » Colleges in Kenya » KASNEB Registration & Results » Secondary Schools Scholarships in Kenya » Undergraduate & Graduate Scholarships for Kenyans
Scholarships for African Students » Undergraduate Scholarships » African Women Scholarships & Grants » Developing Countries Scholarships » Erasmus Mundus Scholarships for Developing Countries » Fellowship Programs » Funding Grants for NGOs » Government Scholarships » LLM Scholarships » MBA Scholarships » PhD and Masters by Research Scholarships » Public Health Scholarships – MPH Scholarships » Refugees Scholarships » Research Grants » Scholarships and Grants
Scholarships in Australia » Scholarships in Belgium » Scholarships in Canada » Scholarships in Germany » Scholarships in Italy » Scholarships in Japan » Scholarships in Korea » Scholarships in Netherlands » Scholarships in UK » Scholarships in USA
Chemistry kcse questions and answer
chemistry paper 3 question and answer
kcse chemistry practical
kcse chemistry paper 2 2016
kcse chemistry paper 1 2016
chemistry paper 2 questions and answers
kcse chemistry notes
kcse chemistry paper 2 2014
kcse chemistry paper 1 2013
“Pdf” Revision Questions Chemistry Form 2
“Pdf” Revision Questions Chemistry Form 3
“Pdf” Revision Questions Chemistry Form 4
“Pdf” Revision Questions Chemistry Form Four
“Pdf” Revision Questions Chemistry Form One
“Pdf” Revision Questions Chemistry Form Three
“Pdf” Revision Questions Chemistry Form Two
1 a a KCSE Past Papers
10th Grade Chemistry Questions and Answers
10th Grade Chemistry Test
11th Ncert Chemistry
12th Class Chemistry Book Free Download
2014 KCSE Marking Schemes
2014 Pdf KCSE Past Papers 2015
2015 Chemistry Essay Questions and Answers Form 4
2016 KCSE Papers
2016 KCSE Prediction Questions
2017 Chemistry Hsc Answers
2017 KCSE Prediction Questions
2018 Chemistry KCSE Leakage
2018 Chemistry KCSE Questions
2018 KCSE Busineness Studies
2018 KCSE Exam
2018 KCSE Leakage
2018 KCSE Prediction Questions
2018 KCSE Questions
2019 Chemistry KCSE Leakage
2019 Chemistry KCSE Questions
2019 KCSE Leakage
2019 KCSE Questions
9th Grade Chemistry Study Guide
A a a Chemistry Notes
a a a Chemistry Notes!
a a a ChemistryNotes!
A a KCSE Past Papers
A Biblical View of Social Justice
A Level Chemistry Biological Molecules Questions
A Level Chemistry Exam Questions by Topic
A Level Chemistry Notes Edexcel
A Level Chemistry Notes Xtremepapers
A Level Chemistry Past Papers
A Level Chemistry Questions and Answers
a Level Chemistry Questions and Answers
A Level Chemistry Questions and Answers (Pdf)
A Level Chemistry Questions and Answers Pdf
A Level Chemistry Questions by Topic Kidney Questions With Markschemes
A Level Chemistry Revision
A Level Chemistry Revision Edexcel
A Level Chemistry Revision Guide
A Level Chemistry Revision Notes
A Level Chemistry Revision Notes Pdf
A Level Chemistry Textbook Pdf
A Level Chemistry Year 1 / as Aqa Exam Questions by Topic
A Level Edexcel Notes a* Chemistry
aa Chemistry Form 3 Questions and Answers
Advance KCSE Past Papers
Advance-africa.com KCSE Rev Quiz
Advantages and Disadvantages.
All Chemistry Essays
All Chemistry Notes for Senior Two
All KCSE Past Papers Chemistry With Making Schemes
All Marking Schemes Questions and Answers
All Past K.c.s.e Questions With Answers
Alliance Mocks 2017
Ap Bio Quizzes
Ap Chemistry 1 Textbook Pdf
Ap Chemistry Essay Questions and Answers
Are Sourced From KNEC.
As Level Chemistry Notes
Atika Chemistry Notes
Atika School Chemistry Notes
B/s Book 2 Notes
Basic Chemistry Books Pdf
basic Chemistry Interview Questions and Answers Pdf
Basic Chemistry Interview Questions and Answers Pdf
Basic Chemistry Pdf
Basic Chemistry Questions and Answers
Basic Chemistry Questions and Answers Pdf
Bbc Bitesize Chemistry Ks3
Bihar Board Chemistry Objective Answer 2017
Bihar Board Chemistry Objective Answer 2018
Bio Answers
Bio Quesions
Chemistry 0478
Chemistry 101
Chemistry 12th
Chemistry 12th Class Notes Pdf
Chemistry 2019 Syllabus
Chemistry All KCSE Short Notes
Chemistry Answers
Chemistry Answers Online Free
Chemistry Answers Quizlet
Chemistry Bk 2 Notes
Chemistry Book 1
Chemistry Book 1 Notes
Chemistry Book 2
Chemistry Book 2 Notes
Chemistry Book 3
Chemistry Book 3 KLB
Chemistry Book 3 Notes
Chemistry Book 4
Chemistry Book 4 Notes
Chemistry Book 4 Pdf
Chemistry Book for Class 11
Chemistry Book Four
Chemistry Book Four Notes
Chemistry Book One
Chemistry Book One Notes
Chemistry Book Pdf Free Download
Chemistry Book Three
Chemistry Book Three Notes
Chemistry Book Three Pdf
Chemistry Book Two
Chemistry Book Two Notes
Chemistry Books Form Three
Chemistry Bowl Chemistry Study Guide
Chemistry Bowl Questions Chemistry
Chemistry Bowl Questions Earth Chemistry
Chemistry Bowl Questions Math
Chemistry Bowl Questions Middle School
Chemistry Brekthrough Form Two Notes
Chemistry Class 12 Ncert Solutions
Chemistry Class 12 Pdf
Chemistry Communication Syllabus
Chemistry Diagram Software
Chemistry Diagrams for Class 11
Chemistry Diagrams for Class 12
Chemistry Diagrams for Class 9
Chemistry Diagrams for Class-10
Chemistry Diagrams in Form 1
Chemistry Diagrams in Form 2
Chemistry Diagrams in Form 3
Chemistry Diagrams in Form 4
Chemistry Diagrams Pdf
Chemistry Diagrams to Label
Chemistry Essay Questions and Answers
Chemistry Essay Questions and Answers 2018
Chemistry Essay Questions and Answers Form 1
Chemistry Essay Questions and Answers Form 2
Chemistry Essay Questions and Answers Form 3
Chemistry Essay Questions and Answers Form 4
Chemistry Essay Questions and Answers Form 4 Pdf
Chemistry Essay Questions and Answers Pdf
Chemistry Essay Revision Q
Chemistry Essays and Answers
Chemistry Essays Form One to Form Four
Chemistry Essays Form One to Form Three
Chemistry Essays KCSE
Chemistry Essays Pdf
Chemistry Exam 1 Multiple Choice
Chemistry Exam 2 Advance
Chemistry Exam 2 Test
Chemistry Exam 2016
Chemistry Exam Form Four
Chemistry Exam Form One
Chemistry Exam Form Three
Chemistry Exam Form Two
Chemistry Exam Practice Test
Chemistry Exam Questions
Chemistry Exam Questions and Answers
Chemistry Exam Questions and Answers Pdf
Chemistry Exam Study Guide
Chemistry Exams
Chemistry Excretion Notes
Chemistry Exercise Form 4 With Answers
Chemistry Final Exam Answer Key
Chemistry Final Exam Answer Key 2016
Chemistry Final Exam Answer Key 2017
Chemistry Final Exam Answers 2018
Chemistry Final Exam Answers 2019
Chemistry Final Exam Questions and Answers
Chemistry Fom 1 Notes
Chemistry Fom 2 Notes
Chemistry Fom 3 Notes
Chemistry Fom 4 Notes
Chemistry Form 1
Chemistry Form 1 & 2 and Answers
Chemistry Form 1 and 2 Essays
Chemistry Form 1 and 2 Essays Questions and Answers
Chemistry Form 1 Chapter 1
Chemistry Form 1 Diagrams
Chemistry Form 1 Exams
Chemistry Form 1 Mid Year Exam
Chemistry Form 1 Notes
Chemistry Form 1 Notes and Questions
Chemistry Form 1 Notes Download
Chemistry Form 1 Notes Free Download
Chemistry Form 1 Notes GCSE
Chemistry Form 1 Notes KCSE-kcse
Chemistry Form 1 Notes Pdf
Chemistry Form 1 Notes Pdf Download
Chemistry Form 1 Past Papers
Chemistry Form 1 Pdf
Chemistry Form 1 Pressure
Chemistry Form 1 Question Papers
Chemistry Form 1 Questions
Chemistry Form 1 Questions and Answers
Chemistry Form 1 Questions and Answers Pdf
Chemistry Form 1 Quiz
Chemistry Form 1 Revision Questions
Chemistry Form 1 Summary Notes
Chemistry Form 1 Syllabus
Chemistry Form 1 Work
Chemistry Form 1-4 Notes
Chemistry Form 2
Chemistry Form 2 Chapter 1
Chemistry Form 2 Chapter 2
Chemistry Form 2 Diagrams
Chemistry Form 2 Exam Paper 2014
Chemistry Form 2 Exams
Chemistry Form 2 Notes
Chemistry Form 2 Notes and Questions
Chemistry Form 2 Notes GCSE
Chemistry Form 2 Notes KCSE-kcse
Chemistry Form 2 Notes Pdf
Chemistry Form 2 Notes Pdf Download
Chemistry Form 2 Past Papers
Chemistry Form 2 Pdf
Chemistry Form 2 Question Papers
Chemistry Form 2 Questions
Chemistry Form 2 Questions and Answers
Chemistry Form 2 Questions and Answers Pdf
Chemistry Form 2 Quiz
Chemistry Form 2 Revision Notes
Chemistry Form 2 Salts
Chemistry Form 2 Structure and Bonding
Chemistry Form 2 Summary Notes
Chemistry Form 2 Syllabus
Chemistry Form 2 Work
Chemistry Form 3
Chemistry Form 3 and 4 Essays
Chemistry Form 3 and 4 Essays Questions and Answers
Chemistry Form 3 Chapter 3
Chemistry Form 3 Classification
Chemistry Form 3 Diagrams
Chemistry Form 3 Ecology
Chemistry Form 3 Exams
Chemistry Form 3 Notes
Chemistry Form 3 Notes and Questions
Chemistry Form 3 Notes GCSE
Chemistry Form 3 Notes KCSE-kcse
Chemistry Form 3 Notes Pdf
Chemistry Form 3 Notes Pdf Download
Chemistry Form 3 Notes Topic 1
Chemistry Form 3 Past Papers
Chemistry Form 3 Pdf
Chemistry Form 3 Question Papers
Chemistry Form 3 Questions
Chemistry Form 3 Questions and Answers
Chemistry Form 3 Questions and Answers Pdf
Chemistry Form 3 Questions and Answers Term 3
Chemistry Form 3 Questions and Answers+pdf
Chemistry Form 3 Quiz
Chemistry Form 3 Revision Notes
Chemistry Form 3 Revision Questions
Chemistry Form 3 Summary Notes
Chemistry Form 3 Syllabus
Chemistry Form 3 Syllabus Pdf
Chemistry Form 3 Topics
Chemistry Form 3 Work
Chemistry Form 4
Chemistry Form 4 All Chapter
Chemistry Form 4 Chapter 1 Conversion of Units
Chemistry Form 4 Chapter 1 Exercise
Chemistry Form 4 Chapter 1 Exercise and Answers
Chemistry Form 4 Chapter 1 Exercise Pdf
Chemistry Form 4 Chapter 1 Mind Map
Chemistry Form 4 Chapter 2
Chemistry Form 4 Chapter 2 Exercise and Answers
Chemistry Form 4 Chapter 2 Exercise Pdf
Chemistry Form 4 Chapter 2 Experiment
Chemistry Form 4 Chapter 2 Formula
Chemistry Form 4 Chapter 2 Mind Map
Chemistry Form 4 Chapter 2 Momentum
Chemistry Form 4 Chapter 2 Notes Pdf
Chemistry Form 4 Chapter 2 Objective Questions and Answers
Chemistry Form 4 Chapter 2 Paper 2
Chemistry Form 4 Chapter 2 Slideshare
Chemistry Form 4 Chapter 3
Chemistry Form 4 Chapter 3 Questions and Answers
Chemistry Form 4 Chapter 4
Chemistry Form 4 Chapter 4 Notes Pdf
Chemistry Form 4 Chapter 5 Light Questions and Answers
Chemistry Form 4 Chapter 5 Notes Pdf
Chemistry Form 4 Diagrams
Chemistry Form 4 Exam Paper 1
Chemistry Form 4 Exams
Chemistry Form 4 Exercise
Chemistry Form 4 Exercise Pdf
Chemistry Form 4 Module With Answer
Chemistry Form 4 Note
Chemistry Form 4 Notes
Chemistry Form 4 Notes (Pdf)
Chemistry Form 4 Notes All Chapter Pdf
Chemistry Form 4 Notes and Questions
Chemistry Form 4 Notes Chapter 1
Chemistry Form 4 Notes Chapter 2
Chemistry Form 4 Notes Chapter 3
Chemistry Form 4 Notes Download
Chemistry Form 4 Notes Free Download
Chemistry Form 4 Notes GCSE
Chemistry Form 4 Notes KCSE-kcse
Chemistry Form 4 Notes Pdf
Chemistry Form 4 Notes Pdf Download
Chemistry Form 4 Paper 2 Questions and Answers
Chemistry Form 4 Past Papers
Chemistry Form 4 Question Papers
Chemistry Form 4 Questions
Chemistry Form 4 Questions and Answers
Chemistry Form 4 Questions and Answers Pdf
Chemistry Form 4 Quiz
Chemistry Form 4 Revision Notes
Chemistry Form 4 Schemes of Work
Chemistry Form 4 Summary Notes
Chemistry Form 4 Syllabus
Chemistry Form 4 Textbook Pdf
Chemistry Form 4 Work
Chemistry Form 5 Chapter 1 Exercise and Answers
Chemistry Form 5 Chapter 1 Notes Pdf
Chemistry Form 5 Chapter 2 Notes Pdf
Chemistry Form 5 Chapter 2 Slideshare
Chemistry Form 5 Chapter 3 Notes Pdf
Chemistry Form 5 Notes Pdf
Chemistry Form Four Book
Chemistry Form Four Notes
Chemistry Form Four Notes and Questions
Chemistry Form Four Notes GCSE
Chemistry Form Four Notes Pdf
Chemistry Form Four Past Papers
Chemistry Form Four Questions
Chemistry Form Four Questions and Answers
Chemistry Form Four Questions and Answers Pdf
Chemistry Form Four Quiz
Chemistry Form Four Study Notes
Chemistry Form Four Syllabus
Chemistry Form Four Topic 2
Chemistry Form Four Topic 4
Chemistry Form Four Topics
Chemistry Form Four Work
Chemistry Form One
Chemistry Form One Book
Chemistry Form One Book Pdf
Chemistry Form One Download Topic 1 Upto 3
Chemistry Form One Exam
Chemistry Form One Notes
Chemistry Form One Notes and Questions
Chemistry Form One Notes GCSE
Chemistry Form One Notes Pdf
Chemistry Form One Pdf
Chemistry Form One Questions
Chemistry Form One Questions and Answers
Chemistry Form One Questions and Answers Pdf
Chemistry Form One Questions and Their Answers
Chemistry Form One Quiz
Chemistry Form One Revision Question
Chemistry Form One Schemes of Work
Chemistry Form One Study Notes
Chemistry Form One Syllabus
Chemistry Form One Term Three Test
Chemistry Form One to Three Notes
Chemistry Form One Work
Chemistry Form Three
Chemistry Form Three Book
Chemistry Form Three Notes
Chemistry Form Three Notes and Questions
Chemistry Form Three Notes GCSE
Chemistry Form Three Questions and Answers
Chemistry Form Three Questions and Answers Pdf
Chemistry Form Three Quiz
Chemistry Form Three Reproduction
Chemistry Form Three Reproduction.
Chemistry Form Three Study Notes
Chemistry Form Three Work
Chemistry Form Three-questions and Answers
Chemistry Form Two
Chemistry Form Two Book
Chemistry Form Two Diagrams
Chemistry Form Two Notes
Chemistry Form Two Notes and Questions
Chemistry Form Two Notes GCSE
Chemistry Form Two Notes Pdf
Chemistry Form Two Notes-pdf
Chemistry Form Two Pdf
Chemistry Form Two Questions
Chemistry Form Two Questions and Answers
Chemistry Form Two Questions and Answers Pdf
Chemistry Form Two Quiz
Chemistry Form Two Study Notes
Chemistry Form Two Topics
Chemistry Form Two Work
Chemistry Form Two,schemes of Work
Chemistry Form2
Chemistry Form2 Textbook
Chemistry Game Form Four Question End Answers
Chemistry Grade 10 Exam Papers
Chemistry Hsc Pdf
Chemistry Human Reproduction Video
Chemistry IGCSE Past Papers Xtremepapers
Chemistry K.c.s.e 2017
Chemistry KCSE
Chemistry KCSE 2016
Chemistry KCSE 2017
Chemistry KCSE 2017 Paper 1
Chemistry KCSE Past Papers
Chemistry KCSE Questions
Chemistry KCSE Questions and Answer
Chemistry KCSE Quizzes & Answers
Chemistry KCSE Revision
Chemistry KCSE Revision Notes
Chemistry KCSE Setting Questions Form One and Two
Chemistry Ksce 2015
Chemistry Last Year K.c.s.e Questions
Chemistry Lesson Plan Form Two
Chemistry Made Familiar
Chemistry Mcq for Class 11
Chemistry Mcq for Class 12
Chemistry Mcq for Competitive Exams
Chemistry Mcq for Competitive Exams Pdf
Chemistry Mcq for Neet Pdf
Chemistry Mcq for Ssc
Chemistry Mcq Questions With Answers
Chemistry Mcq With Answers Pdf
Chemistry Mcqs for Class 12 Pdf
Chemistry Mcqs With Answers Pdf
Chemistry Mid Familia Form One
Chemistry Mock Papers
Chemistry Module Form 5
Chemistry Multiple Choice Questions and Answers Cxc
Chemistry Multiple Choice Questions and Answers Pdf
Chemistry Multiple Choice Questions With Answers Pdf
Chemistry Note
Chemistry Note Form Two All Chapters
Chemistry Notes
Chemistry Notes and Guestion and Answear
Chemistry Notes and Syllabus
Chemistry Notes Class 10
Chemistry Notes for Class 11 Pdf
Chemistry Notes for Class 12 Pdf
Chemistry Notes for High School Students
Chemistry Notes for IGCSE 2014
Chemistry Notes Form 1
Chemistry Notes Form 1 4
Chemistry Notes Form 1 Free Download
Chemistry Notes Form 1 KLB
Chemistry Notes Form 1 Pdf
Chemistry Notes Form 1-4
Chemistry Notes Form 1-4(1) Chemistry
Chemistry Notes Form 14
Chemistry Notes Form 2
Chemistry Notes Form 2 KLB
Chemistry Notes Form 2 Pdf
Chemistry Notes Form 2; Chemistry Notes
Chemistry Notes Form 3
Chemistry Notes Form 3 KLB
Chemistry Notes Form 3 Pdf
Chemistry Notes Form 4
Chemistry Notes Form 4 Chapter 2
Chemistry Notes Form 4 KLB
Chemistry Notes Form 4 Pdf
Chemistry Notes Form 4-pdf
Chemistry Notes Form Four
Chemistry Notes Form Four KLB
Chemistry Notes Form Four Pdf
Chemistry Notes Form One
Chemistry Notes Form One KLB
Chemistry Notes Form One Pdf
Chemistry Notes Form One to Form Four
Chemistry Notes Form Three
Chemistry Notes Form Three KLB
Chemistry Notes Form Three Pdf
Chemistry Notes Form Two
Chemistry Notes Form Two KLB
Chemistry Notes Form Two Pdf
Chemistry Notes Form2
Chemistry Notes IGCSE
Chemistry Notes Kenya
Chemistry Notes on Agroforestry
Chemistry Notes Pdf
Chemistry Notes:
Chemistry Objective Answer
Chemistry Objective Answer 2018
Chemistry Objective Questions for Competitive Exams
Chemistry Objective Questions for Competitive Exams Pdf
Chemistry Oral Exam Questions
Chemistry Paper 1
Chemistry Paper 1 2018 Marking Rules
Chemistry Paper 1 Notes
Chemistry Paper 1 Questions
Chemistry Paper 1 Questions and Answers
Chemistry Paper 1 Topics
Chemistry Paper 1 With Answers
Chemistry Paper 2
Chemistry Paper 2 2017
Chemistry Paper 2 2018 Marking Rules
Chemistry Paper 2 Questions and Answers
Chemistry Paper 2 Questions and Answers Pdf
Chemistry Paper 2 Revision
Chemistry Paper 2 Topics
Chemistry Paper 2018
Chemistry Paper 3 2018 Marking Rules
Chemistry Paper 3 Question and Answer
Chemistry Paper 3 Question Paper 2014 KCSE
Chemistry Paper 3 Question Paper 2015 KCSE
Chemistry Paper 3 Question Paper 2016 KCSE
Chemistry Paper 3 Question Paper 2017 KCSE
Chemistry Paper 3 Question Paper 2018 KCSE
Chemistry Paper 3 Questions and Answers
Chemistry Paper One Questions and Answers
Chemistry Paper One Topics
Chemistry Paper Two Qestions With Answers
Chemistry Paper1
Chemistry Paper2
Chemistry Paper3
Chemistry Paper4
Chemistry Past Papers
Chemistry Past Papers 2017
Chemistry Past Papers a Level
Chemistry Past Papers Form 1
Chemistry Past Papers Form 2
Chemistry Past Papers Form 3
Chemistry Past Papers O Level
Chemistry Pdf Download
Chemistry Pp1 KCSE 2016
Chemistry Practical Book Class 12 Pdf
Chemistry Practical Exam
Chemistry Practicals Form One
Chemistry Practicals Questions and Answers
Chemistry Practice Test 9th Grade
Chemistry Practice Test Answers
Chemistry Practice Test Questions and Answers
Chemistry Practice Test Quizlet
Chemistry Predicted Questions This Year KCSE
Chemistry Preparation Notes
Chemistry Pretest High School Pdf
Chemistry Question and Answer With Explanation
Chemistry Question and Answers 2019
Chemistry Question and Answers 2020
Chemistry Question and Answers 2021
Chemistry Question and Answers 2022
Chemistry Question and Answers Note
Chemistry Questions
Chemistry Questions and Answers
Chemistry Questions and Answers for High School
Chemistry Questions and Answers for High Schools
Chemistry Questions and Answers for High Schools Pdf
Chemistry Questions and Answers for Secondary Schools
Chemistry Questions and Answers Form 1
Chemistry Questions and Answers Form 2
Chemistry Questions and Answers Form 3
Chemistry Questions and Answers Form 4
Chemistry Questions and Answers Multiple Choice
Chemistry Questions and Answers Notes
Chemistry Questions and Answers O
Chemistry Questions and Answers Online
Chemistry Questions and Answers Pdf
Chemistry Questions and Answers Pdf for Class 12
Chemistry Questions and Answers Pdf for Competitive Exams
Chemistry Questions and Answers-form 2
Chemistry Questions for High School
Chemistry Questions for High School Students With Answers
Chemistry Questions for Senior 1
Chemistry Questions for Senior 2
Chemistry Questions for Senior 3
Chemistry Questions for Senior 4
Chemistry Questions for Senior 5
Chemistry Questions for Senior 6
Chemistry Questions for Senior Five
Chemistry Questions for Senior Four
Chemistry Questions for Senior One
Chemistry Questions for Senior Six
Chemistry Questions for Senior Three
Chemistry Questions for Senior Two
Chemistry Questions Form One
Chemistry Questions Multiple Choice
Chemistry Questions Quizlet
Chemistry Questions to Ask Your Teacher
Chemistry Quetion and Answer Form Four
Chemistry Quetion and Answer Form One
Chemistry Quetion and Answer Form Three
Chemistry Quetion and Answer Form Two
Chemistry Quiz for Class 9
Chemistry Quiz for Class 9 Chemistry
Chemistry Quiz Questions and Answers for Class 10
Chemistry Quiz Questions and Answers for Class 10 Pdf
Chemistry Quiz Questions and Answers for Class 12
Chemistry Quiz Questions and Answers for Class 9
Chemistry Quiz Questions and Answers for Class 9 Pdf
Chemistry Quiz Questions and Answers for High School
Chemistry Quiz Questions and Answers Multiple Choice
Chemistry Quiz Questions and Answers Pdf
Chemistry Quiz Questions for Class 12
Chemistry Quiz Questions for College Students
Chemistry Quiz With Answers
Chemistry Quiz With Answers Pdf
Chemistry Quizlet
Chemistry Revision
Chemistry Revision a Level
Chemistry Revision Chemistry Notes Chemistry
Chemistry Revision Exam
Chemistry Revision Examination
Chemistry Revision Form One
Chemistry Revision Notes
Chemistry Revision Notes Chemistry
Chemistry Revision Notes Form 1
Chemistry Revision Notes Form 2
Chemistry Revision Notes Form 3
Chemistry Revision Notes Form 4
Chemistry Revision Notes IGCSE
Chemistry Revision Paper One
Chemistry Revision Questions
Chemistry Revision Questions and Answers
Chemistry Revision Questions and Answers Form 1
Chemistry Revision Questions and Answers Form 2
Chemistry Revision Questions and Answers Form 3
Chemistry Revision Questions and Answers Form 4
Chemistry Revision Questions and Answers Form Four
Chemistry Revision Questions and Answers Form One
Chemistry Revision Questions and Answers Form Three
Chemistry Revision Questions and Answers Form Two
Chemistry Revision Questions Form 1
Chemistry Revision Questions Form 2
Chemistry Revision Questions Form 3
Chemistry Revision Questions Form 4
Chemistry Revision Questions Form Four
Chemistry Revision Questions Form One
Chemistry Revision Questions Form Three
Chemistry Revision Questions Form Two
Chemistry Revision Quiz
Chemistry Revision Test
Chemistry Secondary School Revision
Chemistry Simple Notes
Chemistry Spm Notes Download
Chemistry Spm Notes Pdf
Chemistry Spm Questions
Chemistry Study Form 2
Chemistry Study Guide
Chemistry Study Guide Answer Key
Chemistry Study Guide Answers
Chemistry Study Guide Chemistry Questions and Answers
Chemistry Study Guide Ib
Chemistry Study Guide Pdf
Chemistry Study Guides
Chemistry Study Notes
Chemistry Study Notes Materials Form 1 Pdf
Chemistry Study Notes Materials Form 2 3 Pdf
Chemistry Study Notes Materials Form 2 Pdf
Chemistry Study Notes Materials Form 3 Pdf
Chemistry Study Notes Materials Form 4 Pdf
Chemistry Syllabus in Kenya
Chemistry Syllabus Pdf
Chemistry Test 1 Quizlet
Chemistry Test Questions
Chemistry Test Questions and Answers
Chemistry Test Questions and Answers Pdf
Chemistry Topic One Form Four
Chemistry Topics Form One
Chemistry Unit 1 Quiz
Chemistry Vol 3
Chemistry | Revision Chemistry
Chemistry,form 4
Chemistry.form Four.topic Three
ChemistryExam Form Three
ChemistryModule Form 5
ChemistryNotes
ChemistryNotes for Class 11 Pdf
ChemistryNotes for Class 12 Pdf
ChemistryNotes Form 1
ChemistryNotes Form 1 Free Download
ChemistryNotes Form 2
ChemistryNotes Form 3
ChemistryNotes Form 3 Pdf
ChemistryNotes IGCSE
ChemistryNotes Pdf
ChemistryPast Papers
ChemistryQuestions and Answers Pdf
ChemistrySimple Notes
ChemistrySpm Notes Download
ChemistrySpm Notes Pdf
ChemistrySpm Questions
ChemistryStudy Guide Answers
ChemistryStudy Guide Pdf
ChemistryStudy Guides
Blologytextpapers
Bridge Chemistry
Business Past KCSE Past Papers
Business Studies Form 3 Notes Pdf
Business Studies Form 4 Notes Pdf
C R E Form One KLB
C R E Form One Oli Topic
C.r.e Form 1 Notes Kenya
C.r.e Form 2 Notes Kenya
C.r.e Form 3 Notes
C.r.e Form 3 Notes Kenya
C.r.e Form 3 Pdf
C.r.e Form 4 Notes Kenya
C.r.e Form One Notes Pdf
C.r.e Notes Form 1
C.r.e Revision Notes
C.r.e Short Notes
Cambridge IGCSE Chemistry
Cambridge IGCSE Chemistry 3rd Edition
Cambridge IGCSE Chemistry 3rd Edition Plus Cd South Asia Edition
Cambridge IGCSE Chemistry Answers
Cambridge IGCSE Chemistry Coursebook Pdf Download
Cambridge IGCSE Chemistry Practical Workbook
Cambridge IGCSE Chemistry Revision Guide Pdf
Cambridge IGCSE Chemistry Study and Revision Guide 2nd Edition Pdf
Cambridge IGCSE Chemistry Study and Revision Guide Pdf
Cambridge IGCSE Chemistry Workbook Free Download
Cambridge IGCSE Chemistry Workbook Pdf
Cambridge IGCSE® Chemistry Coursebook
Caucasian Chalk Circle Essay Questions
Chapter 1 Introduction to Chemistry
Chapter 1 Introduction to Chemistry Studies
Cie a Level Chemistry Notes 2016
Cie a Level Chemistry Notes Pdf
Cie Past Papers
Class 10 Chemistry Chapter 1 Mcqs
Class 8 Chemistry Notes KCSE-kcse
College Chemistry Notes
College Chemistry Practice Test
College Chemistry Quiz
College Chemistry Quiz Chapter 1
College Chemistry Quizlet
College Chemistry Study Guide
College Chemistry Study Guide Pdf
College Chemistry Test Questions and Answers
College Chemistry Volume 3 Pdf
College ChemistryNotes
Complete Chemistry for Cambridge IGCSE
Complete Chemistry for Cambridge IGCSE Revision Guide Pdf
County Mocks 2017
Cse Past Papers Chemistry 2017
Dl Chemistry Form 3 Pdf Kusoma
Download Chemistry Form 1
Download Chemistry Form 2
Download Chemistry Form 2 Notes
Download Chemistry Form 3
Download Chemistry Form 3 Notes
Download Chemistry Form 4
Download Chemistry Form Four
Download Chemistry Form One
Download Chemistry Form Three
Download Chemistry Form Two
Download Chemistry Notes Form 3
Download Chemistry Notes Form One
Download ChemistryNotes Form 3
Download Form Three Chemistry Notes
Download Free KCSE Past Papers Chemistry
Download Free KCSE Past Papers From KNEC.
Download KCSE Past Papers With Answers
Download KCSE Revision Notes
Download KLB Chemistry Book 2
Download KLB Chemistry Book 3
Download KLB Chemistry Book 4
Download Notes of Chemistry
Downloads | Chemistry | Form Four Exams | Exams
Downloads | Chemistry | Form One Exams | Exams
Downloads | Chemistry | Form Three Exams | Exams
Downloads | Chemistry | Form Two Exams | Exams
Downloads | KCSE Papers and Marking Schemes |
Dvance KCSE Past Papers
Easy Chemistry Questions
Edexcel a Level Chemistry B
Edexcel a Level Chemistry Notes Pdf
Edexcel a Level Chemistry Salters Nuffield
Edexcel A2 Chemistry Notes
Edexcel as Chemistry Revision Guide Pdf
Edexcel Chemistry A2 Revision Notes Pdf
Edexcel Chemistry Unit 2 Revision Notes
Edexcel GCSE Chemistry Revision Guide Pdf
Edexcel IGCSE Chemistry Past Papers
Edexcel IGCSE Chemistry Revision Guide Free Pdf Download
Edexcel IGCSE Chemistry Revision Guide Pdf
Edexcel IGCSE Chemistry Revision Guide Pdf Download
Electronics Form Four Notes
Energy Questions Chemistry Bowl
Essay Questions and Answers KCSE Chemistry Notes
Essay Questions and Answers on Betrayal in the City
Essay Questions Based on Betrayal in the City
Evolving World Chemistry Book 1 Pdf
Evolving World Chemistry Book 4 Notes
Evolving World Chemistry Book Form 1
Evolving World-history Book 3
Exam Notes for Chemistry 101
Exams KCSE Chemistry Paper 1 Questions and Answers
F3 Chemistry Test Paper
Find Download KCSE Past Papers With Answers
Find KCSE Chemistry Essay Questions and Answers
Form 1 Chemistry Exam
Form 1 Chemistry Notes
Form 1 Chemistry Questions and Answers
Form 1 Chemistry Questions and Answers Pdf
Form 1 Chemistry Revision Notes
Form 1 Chemistry Summurized Revision Pdf
Form 1 Chemistry Syllabus
Form 1 Chemistry Test Paper Pdf
Form 1 Chemistry Topics
Form 1 ChemistryNotes
Form 1 ChemistryQuestions and Answers
Form 1 ChemistryRevision Notes
Form 1 ChemistrySyllabus
Form 1 ChemistryTest Paper Pdf
Form 1 Past Papers
Form 1 Past Papers With Answers
Form 1 Revision Papers
Form 1 Subjects in Kenya
Form 2 Chemistry Exam
Form 2 Chemistry Exam Paper
Form 2 Chemistry Exam Paper 2016
Form 2 Chemistry Exam Paper Free Download
Form 2 Chemistry Exam Paper With Answer
Form 2 Chemistry Final Year Exam Paper 2
Form 2 Chemistry Notes
Form 2 Chemistry Notes and Revision Questions
Form 2 Chemistry Notes Pdf
Form 2 Chemistry Past Papers
Form 2 Chemistry Questions
Form 2 Chemistry Questions and Answers
Form 2 Chemistry Questions and Answers >
Form 2 Chemistry Questions and Answers Pdf
Form 2 Chemistry Revision Notes
Form 2 Chemistry Short Notes
Form 2 Chemistry Syllabus
Form 2 ChemistryExam Paper
Form 2 ChemistryExam Paper Free Download
Form 2 ChemistryExam Paper With Answer
Form 2 ChemistryFinal Year Exam Paper 2
Form 2 ChemistryPast Papers
Form 2 ChemistryRevision Notes
Form 2 ChemistryShort Notes
Form 2 ChemistrySyllabus
Form 2 Revision Papers
Form 2 Subjects in Kenya
Form 3 Chemistry Book
Form 3 Chemistry Exam
Form 3 Chemistry Exam Paper
Form 3 Chemistry Notes
Form 3 Chemistry Past Papers
Form 3 Chemistry Questions
Form 3 Chemistry Questions and Answers
Form 3 Chemistry Questions and Answers Pdf
Form 3 Chemistry Revision Notes
Form 3 Chemistry Syllabus
Form 3 ChemistryExam Paper
Form 3 ChemistryNotes
Form 3 ChemistryPast Papers
Form 3 ChemistryQuestions
Form 3 ChemistryQuestions and Answers Pdf
Form 3 ChemistryRevision Notes
Form 3 ChemistrySyllabus
Form 3 C.r.e
Form 3 Notes of Chemistry Topic on Fish
Form 3 Past Papers
Form 3 Revision Papers
Form 3 Subjects in Kenya
Form 4 Chemistry Exam
Form 4 Chemistry Notes
Form 4 Chemistry Notes Pdf
Form 4 Chemistry Questions and Answers
Form 4 Chemistry Questions and Answers Pdf
Form 4 Chemistry Revision Notes
Form 4 Chemistry Syllabus
Form 4 Chemistry Topics
Form 4 ChemistryNotes
Form 4 ChemistryRevision Notes
Form 4 ChemistrySyllabus
Form 4 ChemistryTopics
Form 4 Exam Papers
Form 4 Revision Papers
Form 4 Subjects in Kenya
Form 5 Chemistry Topics
Form 5 ChemistryTopics
Form Five Chemistry Notes
Form Five ChemistryNotes
Form Four Chemistry Book
Form Four Chemistry Notes
Form Four Chemistry Notes Pdf
Form Four Chemistry Questions and Answers
Form Four Chemistry Questions and Answers Pdf
Form Four Chemistry Revision Questions
Form Four Chemistry Syllabus
Form Four Chemistry Topics
Form Four ChemistryNotes
Form Four ChemistryQuestions and Answers
Form Four ChemistryQuestions and Answers Pdf
Form Four ChemistryTopics
Form Four Notes
Form Four Revision Papers
Form Four Subjects in Kenya
Form One Chemistry Book
Form One Chemistry Examination
Form One Chemistry First Topic
Form One Chemistry Lesson Plan
Form One Chemistry Notes Pdf
Form One Chemistry Past Papers Pdf
Form One Chemistry Questions
Form One Chemistry Questions and Answers
Form One Chemistry Questions and Answers Pdf
Form One Chemistry Revision Questions
Form One Chemistry Short Notes
Form One Chemistry Syllabus
Form One Chemistry Topics
Form One ChemistryExamination
Form One ChemistryPast Papers Pdf
Form One ChemistryQuestions and Answers
Form One ChemistryQuestions and Answers Pdf
Form One ChemistryTopics
Form One Exams
Form One Notes of Chemistry
Form One Past Papers
Form One Subjects in Kenya
Form One Term One Chemistry Exam
Form One Term One ChemistryExam
Form Three Chemistry Book
Form Three Chemistry Book Pdf
Form Three Chemistry Notes
Form Three Chemistry Notes Pdf
Form Three Chemistry Questions and Answers
Form Three Chemistry Questions and Answers Pdf
Form Three Chemistry Revision Questions
Form Three Chemistry Syllabus
Form Three Chemistry Topics
Form Three ChemistryNotes
Form Three ChemistryNotes Pdf
Form Three ChemistryQuestions and Answers
Form Three ChemistryQuestions and Answers Pdf
Form Three ChemistryTopics
Form Three Subjects in Kenya
Form Two Chemistry Book
Form Two Chemistry Cat
Form Two Chemistry Examination
Form Two Chemistry Notes
Form Two Chemistry Notes Pdf
Form Two Chemistry Past Papers
Form Two Chemistry Questions and Answers
Form Two Chemistry Questions and Answers Pdf
Form Two Chemistry Revision Questions
Form Two Chemistry Syllabus
Form Two Chemistry Topics
Form Two ChemistryNotes
Form Two ChemistryNotes Pdf
Form Two ChemistryQuestions and Answers
Form Two ChemistryQuestions and Answers Pdf
Form Two ChemistrySyllabus
Form Two ChemistryTopics
Form Two Notes
Form Two Subjects in Kenya
Free a-level Chemistry Revision App | Pass Your Chemistry Exams
Free Chemistry Form 1 Notes
Free Chemistry Notes Form 1
Free Chemistry Notes Pdf
Free ChemistryNotes Pdf
Free College Chemistry Practice Test
Free Form1,form2,form3 Past Papers Free KCSE Past Papers
Free KCSE Mocks 2015
Free KCSE Past Papers 2014
Free KCSE Past Papers KCSE Past
Free KCSE Past Papers Kenya,
Free KCSE Past Papers With Answers
Free KCSE Questions and Answers on Chemistry
Free KCSE Revision Notes
Free Marking Schemes
Free Mocks Online KCSE Answers Past Exams Question Papers
Free Revision Papers
From Three Notes Topic One KLB
Fun Chemistry Questions
Funny Chemistry Questions
Funny Chemistry Questions and Answers
Funny Chemistry Questions to Ask
Funny Chemistry Quotes
GCSE Chemistry Exam Questions and Answers
GCSE Chemistry Past Papers
GCSE Chemistry Revision
GCSE Chemistry Revision Notes
GCSE Chemistry Revision Notes Pdf
GCSE Chemistry Revision Notes Pdf 9-1
GCSE Chemistry Revision Questions and Answers
GCSE Chemistry Textbook Pdf
GCSE Chemistry Topics Pass My Exams: Easy Exam Revision Notes
General Chemistry Notes Pdf
General Chemistry Practice Test With Answers
General Chemistry Quiz
General Chemistry Quiz Pdf
General Chemistry Test Questions and Answers
General Chemistry Test Questions and Answers Pdf
General Knowledge in Chemistry Human Body
Good Chemistry Questions to Ask
GRE Chemistry Practice Test
GRE Chemistry Subject Test Pdf
Handbook of Chemistry Pdf Free Download
Hard Chemistry Questions
Hard Chemistry Questions and Answers
Hard Chemistry Questions to Ask Your Teacher
Hard Chemistry Quiz Questions
Hard Form 3 Chemistry Question
High School Chemistry Final Exam Doc
High School Chemistry Final Exam Pdf
High School Chemistry Final Exam Questions
High School Chemistry Final Exam Questions and Answers
High School Chemistry Notes
High School Chemistry Practice Test
High School Chemistry Pretest With Answers
High School Chemistry Questions and Answers Pdf
High School Chemistry Study Guide
High School Chemistry Test Questions and Answers Pdf
High School ChemistryNotes
High School ChemistryStudy Guide
How to Answer KCSE Chemistry Question
How to Motivate a Form 4 Student
How to Motivate a KCSE Candidate
How to Motivate a KCSE Student
How to Pass Chemistry Questions & Answers Form 1&2 | Text Book
How to Revise Chemistry
How to Revise Effectively for KCSE
How to Study Chemistry: 5 Study Techniques to Master Chemistry
Hsc Chemistry 2018
Hsc Chemistry 2019
Https://www.knec.ac.ke/ Www.knec-portal.ac.ke/ KNEC Portal:
Ial Chemistry Notes
Ib Chemistry Cold War Notes
Ib Chemistry Notes
Ib Chemistry Notes Pdf
Ib Chemistry of the Americas Notes
Ib Chemistry of the Americas Study Guide
Ib Chemistry Paper 2 Study Guide
Ib Chemistry Question Bank by Topic
Ib Chemistry Study Guide Pdf
Ict Notes Form 1
IGCSE Chemistry Alternative to Practical Revision
IGCSE Chemistry Alternative to Practical Revision Notes
IGCSE Chemistry Book
IGCSE Chemistry Book Pdf Download
IGCSE Chemistry Notes
IGCSE Chemistry Notes 2017 Pdf
IGCSE Chemistry Notes Edexcel
IGCSE Chemistry Paper 2 Notes
IGCSE Chemistry Paper 6 Notes
IGCSE Chemistry Past Papers
IGCSE Chemistry Past Papers 2014
IGCSE Chemistry Past Papers 2017
IGCSE Chemistry Pdf
IGCSE Chemistry Pre Release Material 2018
IGCSE Chemistry Resources
IGCSE Chemistry Revision Guide
IGCSE Chemistry Revision Guide Free Download
IGCSE Chemistry Revision Guide Pdf Download
IGCSE Chemistry Revision Notes Pdf
IGCSE Chemistry Revision Worksheets
IGCSE Chemistry Workbook Pdf
IGCSE Chemistry Znotes
IGCSE ChemistryPast Papers
IGCSE Notes Chemistry
Importance of Agroforestry
Inorganic Chemistry Multiple Choice Questions With Answers Pdf
Inorganic Chemistry Questions and Answers Pdf
Interesting Chemistry Questions
Interesting Chemistry Questions and Answers
Interesting Questions to Ask About Chemistry
Intro to Chemistry Quiz
Introduction of Chemistry Form One
Introduction to Chemistry
Introduction to Chemistry Notes
Introduction to Chemistry Pdf
Introduction to ChemistryNotes
Is Agroforestry Sustainable?
K.c.s.e Answers Chemistry Paper One 2018
K.c.s.e Chemistry 2017
K.c.s.e Chemistry 2018
K.c.s.e Chemistry Paper 1 2017
K.c.s.e Mocks 2018
K.c.s.e Papers 2015
K.c.s.e Papers 2016
K.c.s.e Past Papers 2014
K.c.s.e.Chemistry Paper 2 Year 2018
K.c.s.e.results 2018 for Busia County
K.l.b Chemistry Form 3
K.l.b Chemistry Notes
K.l.b ChemistryNotes
Kasneb Past Papers for Colleges Chemistry Past Papers
KCSE 2010 Marking Scheme
KCSE 2010 Past Papers
KCSE 2011 Chemistry Paper 1
KCSE 2011 Marking Scheme
KCSE 2012 Chemistry Paper 2 Marking Scheme
KCSE 2012 Marking Schemes
KCSE 2013 Chemistry Paper 1
KCSE 2013 Marking Scheme
KCSE 2013 Marking Scheme Pdf
KCSE 2014
KCSE 2015 Chemistry Paper 2
KCSE 2015 Chemistry Paper 3
KCSE 2015 Marking Scheme
KCSE 2015 Past Papers
KCSE 2016 Chemistry Paper 1
KCSE 2016 Chemistry Paper 2
KCSE 2017 Chemistry Paper 1
KCSE 2017 Chemistry Paper 2
KCSE 2017 Hostory Papers With Answers.com
KCSE 2017 Marking Scheme
KCSE 2017 Papers
KCSE 2017 Papers and Marking Scheme
KCSE 2017 Papers Pdf
KCSE 2017 Past Papers
KCSE 2017 Prediction Pdf
KCSE 2018 Chemistry and Answers
KCSE 2018 Chemistry Prediction
KCSE 2018 Leakage
KCSE 2018 Marking Scheme
KCSE 2018 Papers
KCSE 2018 Prediction Pdf
KCSE 2018 Predictions
KCSE 2018 Questions
KCSE 2018 Questions and Answers
KCSE 2019 Leakage Chemistry
KCSE 2019 Marking Scheme
KCSE 2019 Questions
KCSE 2019 Questions and Answers
KCSE 2020 Questions
KCSE 2020 Questions and Answers
KCSE Answers
KCSE Answers Past Exams Question Papers Downloads |
KCSE Chemistry 2011
KCSE Chemistry 2016
KCSE Chemistry Diagramsbiology Revision Tips
KCSE Chemistry Essay Questions and Answers
KCSE Chemistry Essay Questions and Answers Pdf
KCSE Chemistry Essays
KCSE Chemistry Essays Pdf
KCSE Chemistry Marking Schemes
KCSE Chemistry Notes
KCSE Chemistry Notes Pdf
KCSE Chemistry Notes, Syllabus, Questions, Answers
KCSE Chemistry Paper 1
KCSE Chemistry Paper 1 2011
KCSE Chemistry Paper 1 2012
KCSE Chemistry Paper 1 2013
KCSE Chemistry Paper 1 2015
KCSE Chemistry Paper 1 2016
KCSE Chemistry Paper 1 2017
KCSE Chemistry Paper 1 2017 Pdf
KCSE Chemistry Paper 1 Questions and Answers
KCSE Chemistry Paper 2
KCSE Chemistry Paper 2 2012
KCSE Chemistry Paper 2 2012 KCSE Chemistry Paper 2 2015
KCSE Chemistry Paper 2 2013
KCSE Chemistry Paper 2 2014
KCSE Chemistry Paper 2 2015
KCSE Chemistry Paper 2 2016
KCSE Chemistry Paper 2 2017
KCSE Chemistry Paper 3
KCSE Chemistry Paper 3 2012
KCSE Chemistry Paper 3 2016
KCSE Chemistry Paper 3 2017
KCSE Chemistry Paper 3 Past Papers
KCSE Chemistry Past Papers
KCSE Chemistry Past Papers and Answers
KCSE Chemistry Past Papers Pdf
KCSE Chemistry Practical
KCSE Chemistry Practical 2015
KCSE Chemistry Practical 2016
KCSE Chemistry Practical Past Papers
KCSE Chemistry Practicals
KCSE Chemistry Practicals KCSE Chemistry Paper 1
KCSE Chemistry Question and Answer
KCSE Chemistry Questions and Answers
KCSE Chemistry Questions and Answers Ap Chemistry
KCSE Chemistry Revision
KCSE Chemistry Revision Notes
KCSE Chemistry Revision Papers
KCSE Chemistry Revision Questions
KCSE Chemistry Revision Questions and Answers
KCSE Chemistry Syllabus
KCSE ChemistryNotes
KCSE ChemistryPaper 1
KCSE ChemistryPaper 2
KCSE ChemistryPaper 2 Pdf
KCSE ChemistrySyllabus
KCSE Business Paper 1 2016
KCSE Business Past Papers
KCSE Business Studies Past Papers
KCSE Essay Questions in Betrayal in the City
KCSE Essays
KCSE Exam Papers 2018
KCSE Exam Papers Answers
KCSE Form 1 Chemistry Revision
KCSE Form 2 Chemistry Revision
KCSE Form 3 Chemistry Revision
KCSE Form 4 Chemistry Revision
KCSE Form Four Chemistry Revision
KCSE Form One Chemistry Revision
KCSE Form Three Chemistry Revision
KCSE Form Two Chemistry Revision
KCSE KCSE Past Papers KNEC
KCSE Leakage
KCSE Leakage Chemistry
KCSE Made Familiar Chemistry
KCSE Made Familiar Chemistry Pdf
KCSE Marking Scheme 2016
KCSE Marking Schemes
KCSE Marking Schemes 2017
KCSE Marking Schemes Pdf
KCSE Mock Exams
KCSE Mock Papers 2015
KCSE Mock Papers 2017
KCSE Mock Papers 2018
KCSE Mock Papers Pdf
KCSE Mock Papers Pdf 2018
KCSE Mock Papers Pdf KCSE Past Papers
KCSE Mocks 2017
KCSE Mocks 2018
KCSE Notes
KCSE Online Notes
KCSE Online Past Papers
KCSE Online Registration
KCSE Papers 2015
KCSE Papers and Marking Schemes | Exams
KCSE Past Papers
KCSE Past Papers 2007
KCSE Past Papers 2009
KCSE Past Papers 2010
KCSE Past Papers 2011
KCSE Past Papers 2011 Pdf
KCSE Past Papers 2012
KCSE Past Papers 2013
KCSE Past Papers 2013knec
KCSE Past Papers 2014
KCSE Past Papers 2014 Pdf
KCSE Past Papers 2015
KCSE Past Papers 2015 Marking Schemes
KCSE Past Papers 2015 Pdf
KCSE Past Papers 2016
KCSE Past Papers 2016 Pdf
KCSE Past Papers 2017
KCSE Past Papers 2017 Pdf
KCSE Past Papers 2018
KCSE Past Papers Chemistry
KCSE Past Papers Chemistry and Answers
KCSE Past Papers Chemistry Pdf
KCSE Past Papers Chemistry With Answers
KCSE Past Papers Chemistryand Answers
KCSE Past Papers Business Studies and Answers
KCSE Past Papers KCSE and Answers
KCSE Past Papers KCSE and Answers Free Mocks Online
KCSE Past Papers Marking Scheme
KCSE Past Papers Pdf Download
KCSE Past Papers Pdf Download KCSE 2013
KCSE Past Papers With Answers
KCSE Past Papers Woodwork and Answers
KCSE Prediction 2017
KCSE Prediction 2018
KCSE Prediction 2018 Pdf
KCSE Prediction Papers 2018
KCSE Prediction Questions
KCSE Prediction Questions 2018
KCSE Prediction Questions and Answers
KCSE Questions
KCSE Questions and Answers
KCSE Questions and Answers.
KCSE Questions on Chemistry
KCSE Results, Online Registration, KCSE Result Slip.
KCSE Revision
KCSE Revision Notes
KCSE Revision Notes Chemistry
KCSE Revision Notes Pdf
KCSE Revision Papers
KCSE Revision Papers 2014
KCSE Revision Papers With Answers
KCSE Revision Question for Chemistry
KCSE Revision Questions
KCSE Revision Questions and Answers
KCSE Revision | Secondary School | Text Books | Text Book Centre
KCSE Syllabus Pdf
KCSE Trial 2017
KCSE Trial Exams 2017
Kenya Secondary School Chemistry Syllabus
Kenya Secondary School Chemistry Syllabus Pdf
Kenya Secondary School ChemistrySyllabus Pdf
Kenya Secondary School Syllabus Pdf
Kenya-kcse-christian Religious Education Syllabus
Kenyaplex KCSE Past Papers
Kenyaplex Past Papers for Secondary
KLB Chemistry Book 1 Download
KLB Chemistry Book 1 Notes
KLB Chemistry Book 1 Pdf
KLB Chemistry Book 2
KLB Chemistry Book 2 Notes
KLB Chemistry Book 2 Notes Pdf
KLB Chemistry Book 2 Pdf
KLB Chemistry Book 3 Notes
KLB Chemistry Book 3 Pdf
KLB Chemistry Book 3 Pdf Download
KLB Chemistry Book 4 Notes
KLB Chemistry Book 4 Pdf
KLB Chemistry Book 4 Pdf Download
KLB Chemistry Book 4 Topics
KLB Chemistry Book One
KLB Chemistry Form 1
KLB Chemistry Form 1 Notes
KLB Chemistry Form 1 Pdf
KLB Chemistry Form 2
KLB Chemistry Form 2 Book
KLB Chemistry Form 2 Notes
KLB Chemistry Form 2 Pdf
KLB Chemistry Form 2 Pdf Download
KLB Chemistry Form 2 Schemes of Work
KLB Chemistry Form 3
KLB Chemistry Form 3 Notes
KLB Chemistry Form 3 Notes Pdf
KLB Chemistry Form 3 Pdf
KLB Chemistry Form 3 Pdf Download
KLB Chemistry Form 4
KLB Chemistry Form 4 Notes
KLB Chemistry Form 4 Pdf
KLB Chemistry Form Four
KLB Chemistry Form Four Notes
KLB Chemistry Form One
KLB Chemistry Form One Notes
KLB Chemistry Form Three
KLB Chemistry Form Three Notes
KLB Chemistry Form Two
KLB Chemistry Form Two Notes
KLB Chemistry Notes
KLB Chemistry Notes Form 4
KLB Chemistry Pdf
KLB ChemistryNotes
KLB ChemistryNotes Form 4
KLB ChemistryPdf
KNEC Chemistry Syllabus
KNEC Examiners Portal KNEC Website
KNEC Ict Past Papers
KNEC Past Papers for Colleges
KNEC Past Papers Free Download
KNEC Past Papers Free Downloads
KNEC Past Papers Pdf
KNEC Portal Confirmation
KNEC Portal KCSE Results
KNEC Portal KNEC Past Papers for Colleges Kasneb Past Papers
KNEC Revision Papers
KNEC Technical Exams Past Papers
Kusoma Chemistry Notes
Kusoma Chemistry Notes Pdf
Kusoma Notes Chemistry
Kusoma.co.ke
Kusoma.com Past Papers
Learner Guide for Cambridge IGCSE Chemistry
Longhorn Chemistry Book 3 Pdf
Made Familiar Chemistry
Made Familiar Chemistry Pdf
Made Familiar Chemistry Questions
Maktaba Tetea Notes
Marking Scheme KCSE Chemistry Past Papers
Math Form2 Note
Mcqs About Gaseous Exchange
Middle School Chemistry Bowl Chemistry Questions
Mock Past Papers 2017
Mock Past Papers With Answers
Mokasa Mock 2017
More Than 1800 Chemistry Questions and Answers to Help You Study
Multiple Choice Questions on Chemistry
Necta Chemistry Past Papers
Necta Chemistry Practicals
Necta ChemistryPast Papers
Necta ChemistryPracticals
Necta Form Four Past Papers
Necta Past Papers Form 4
Necta Past Papers Form 4 2016
Necta Past Papers Form Six
Necta Past Papers Form Two
Necta Questions and Answers
Necta Review Questions
Notes Chemistry Form 1
Notes Chemistry Form 2
Notes Chemistry Form 3
Notes Chemistry Form 3 Notes Pdf
Notes Chemistry Form 3 Syllabus
Notes Chemistry Form 4 Syllabus
Notes on Chemistry Studies
Notes Za Chemistry 4m 2
Notes Za Chemistry Form One
Notes Za Chemistry Form Three
O Level Chemistry Practical Experiments
O Level Chemistry Questions and Answers Pdf
Orm Three Chemistry Notes
Page Navigation
Papacambridge Chemistry IGCSE
Papers KNEC KCSE Online Past
Papers KNEC KCSE Results Past Papers
Past KCSE Papers
Past Paper Questions by Topic Chemistry
Past Papers 2014
Past Papers in Kenya
Pdf Chemistry Form 3
Pdf Chemistry Notes
Pdf Chemistry Notes Form 1
Pdf Chemistry Notes Form 2
Pdf Chemistry Notes Form 3
Pdf Chemistry Notes Form 4
Pdf Chemistry Notes Form Four
Pdf Chemistry Notes Form One
Pdf Chemistry Notes Form Three
Pdf Chemistry Notes Form Two
Pdf Form 1 Chemistry Questions and Answers
Pdf Form 2 Chemistry Questions and Answers
Pdf Form 3 Chemistry Questions and Answers
Pdf Form 4 Chemistry Questions and Answers
Pdf Form Four Chemistry Questions and Answers
Pdf Form One Chemistry Questions and Answers
Pdf Form Three Chemistry Questions and Answers
Pdf Form Two Chemistry Questions and Answers
Pdf Free KCSE Past Papers and Marking Schemes
Pdf” Revision Questions Chemistry Form 1
Practical Chemistry Experiments Pdf
Practical Chemistry Question and Answer Pdf
Pre Mocks 2018
Preliminary Chemistry
Primary and Secondary Tillage Implements Ppt
Pte KNEC Past Papers
Questions and Answers Pdf Chemistry Form 1
Questions and Answers Pdf Chemistry Form 2
Questions and Answers Pdf Chemistry Form 3
Questions and Answers Pdf Chemistry Form 4
Questions and Answers Pdf Chemistry Form Four
Questions and Answers Pdf Chemistry Form One
Questions and Answers Pdf Chemistry Form Three
Questions and Answers Pdf Chemistry Form Two
Questions Based to Introduction to Chemistry
Questions on Gaseous Exchange in Humans
Questions on Introduction to Chemistry
Questions to Ask in Chemistry Class
Questions to Confuse Your Chemistry Teacher
Quizlet Chemistry Test
Quizlet Test Questions
Qustions in Chemistry and Answers
Revision
Revision Chemistry Notes and Questions?
Revision Quiz for Chemistry for Form Three
S.1 Chemistry Questions
S.2 Chemistry Questions
S.3 Chemistry Questions
S.4 Chemistry Questions
Sample Essays on Betrayal in the City
School Chemistry Notes
Secondary Chemistry Notes
Secondary Chemistry Notes Pdf
Secondary ChemistryNotes Pdf
Senior 1 Chemistry Notes
Senior 2 Chemistry Notes
Senior 3 Chemistry Notes
Senior 4 Chemistry Notes
Senior 5 Chemistry Notes
Senior 6 Chemistry Notes
Senior Five Chemistry Notes
Senior Four Chemistry Notes
Senior One Chemistry Notes
Senior Six Chemistry Notes
Senior Three Chemistry Notes
Senior Two Chemistry Notes
Simple Scientific Questions
Smart Questions to Ask a Chemistry Teacher
Snab Chemistry Revision Notes
Southwest Mock Paper 2 2016 Chemistry Only
Spm Chemistry Revision Notes
Spm Notes
Success Chemistry Spm Pdf
Success ChemistrySpm Pdf
Summary of Chemistry Form 3
Tahossa Past Papers
To Motivate a Form 4 KCSE Student
To Motivate a Form 4 Student
Topical Revision Material
Tricky Chemistry Questions and Answers
Tricky Chemistry Questions for Adults
Tricky Chemistry Questions With Answers
Tricky Chemistry Quiz Questions
Two Chemistry Revision Questions
University Chemistry Volume 3 Openstax
University Chemistry Volume 3 Pdf
University Chemistry Volume 4 Pdf
Ur Revision Guide IGCSE Chemistry
What Are the Types of Gametes
Working of Excretory System
Www.Chemistry Form One Notes.com
Www.Chemistry From One KLB.com
Www.form 1 Chemistry.com
Www.form 2 Chemistry.com
Www.form 3 Chemistry.com
Www.form 4 Chemistry.com
Www.form Four Chemistry.com
Www.form One Chemistry.com
Www.form Three Chemistry.com
Www.form Two Chemistry.com
Www.kusoma Notes
Www.kusoma Revision Materials
Www.kusoma.co.ke Chemistry Notes
Xtremepapers IGCSE Chemistry
Year 11 Chemistry
Z Notes Chemistry IGCSE
Znotes as Chemistry
[ad_2]
Source link
Comments
The Kenyan Digest Team

You may like
National Youth Service Officially Launches 2026 Nationwide Recruitment Exercise for Kenyan Youth
UDA Politician Arrested Over Sixty Million Shilling Fake Tender Scam at the Office of the President
UDA Politician Arrested Over Sixty Million Shilling Fake Tender Scam at the Office of the President
National Youth Service Officially Launches 2026 Nationwide Recruitment Exercise for Kenyan Youth

Xiaomi Unveils REDMI Note 15 Series: REDMI Titan Durability Meets Advanced Imaging

SportyBet’s 1UP Early Win Is a Game-Changer for Kenyan Punters in 2026

Evidence-Based Study Strategies for Academic Success in Higher Education

Securing Scholarships for International Study: A Comprehensive Guide for Students

Paul Ndung’u Bankruptcy Exposes a Stunning Fall After SportPesa Court Defeat

Arsenal vs Manchester United Prediction: Betting Tips, Odds, and Match Preview

EXPOSED: Court Filings Show How Cs Mbadi, Watanga and Nyawanda Approved Sh5.5 Billion Illegal Rice Imports
